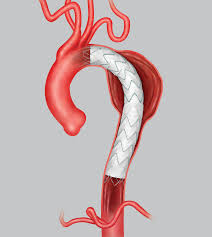
IRVINE, Calif.–(BUSINESS WIRE)–Endologix, Inc. (NASDAQ:ELGX), a developer and marketer of innovative treatments for aortic disorders, today announced positive interim results from the LEOPARD (Looking at Evar Outcomes by Primary Analysis of Randomized Data) clinical study. LEOPARD is the first and only head-to-head, prospective, multi-center, randomized clinical study comparing currently available endovascular abdominal aortic stent grafts. LEOPARD […]
Other News
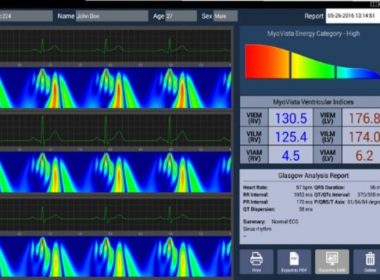
HeartSciences Announces CE Mark and European Launch of MyoVista® High Sensitivity ECG Device
WESTLAKE, Texas, Aug. 17, 2017 /PRNewswire/ — HeartSciences today announced the European launch of MyoVista® high sensitivity electrocardiograph (hsECG™) Testing Device, developed in response to the global unmet need for effective, low-cost, front-line screening of cardiac disease in both symptomatic and asymptomatic patients. MyoVista measures the heart’s energy during each […]
Minnesota Supreme Court allows shareholder suit over Medtronic-Covidien merger
Updated to clarify that MDT officers and directors were compensated for excise-tax based liabilities related to stock options, and not for capital gains. Medtronic (NYSE:MDT) shareholders seeking to sue the company claiming they were harmed by its $50 billion corporate inversion merger with Covidien will get their chance in court, the […]
Cook has Class I Recall for Zenith Alpha Thoracic Graft
Cook said to be recalling this device because of blood clots that can form within the graft after implantation. Zenith Alpha Thoracic Endovascular Graft by Cook Medical: Class I Recall – Potential Formation of Thrombus Inside Device AUDIENCE: Risk Manager, Cardiology, Surgery, Patient ISSUE: Cook Medical Inc. is recalling the […]
BioStable Science & Engineering Announces FDA Clearance of the HAART™ 200 Aortic Annuloplasty Device for Bicuspid Aortic Valve Repair
AUSTIN, Texas–(BUSINESS WIRE)–BioStable Science & Engineering, Inc. announced today it has received FDA market clearance for the HAART 200 Aortic Annuloplasty Device, the first and only annuloplasty device designed specifically for bicuspid aortic valve repair. With FDA clearance of both the HAART 300 and HAART 200 Aortic Annuloplasty Devices, BioStable […]
VentureMed Group Raises $15 Million Series B Equity Financing
TOLEDO, Ohio, Aug. 15, 2017 /PRNewswire/ — VentureMed Group®, Inc., (“VMG” or the “Company”) a medical device company developing and commercializing next-generation endovascular products to treat patients suffering from peripheral artery disease (PAD), today announced it had raised $15 million in new equity financing. The Series B Round was led by new investor Endeavour […]
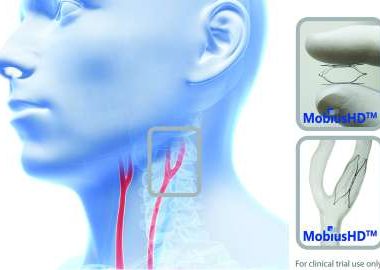
Vascular Dynamics, Inc. Announces FDA Approval To Initiate CALM 2 Trial For Mobiushd System For The Treatment Of Resistant Hypertension
MOUNTAIN VIEW, Calif., Aug. 15, 2017 (GLOBE NEWSWIRE) — Vascular Dynamics, Inc., (VDI) a privately held medical device company developing novel solutions for the treatment of hypertension, today announces that the United States Food and Drug Administration (FDA) has approved the company’s Investigational Device Exemption (IDE) application to initiate its […]
Medtronic Announces Randomized Global Resolute Onyx(TM) DES One-Month Dual Antiplatelet Therapy Study to Address Critical Unanswered Question in Interventional Cardiology
DUBLIN- August 14, 2017 – Medtronic plc (NYSE: MDT) today announced a global randomized clinical trial that will evaluate one-month dual antiplatelet therapy (DAPT) – the combination of aspirin and an anti-clotting medication – in patients implanted with the Resolute Onyx(TM) Drug-Eluting Stent (DES) during percutaneous coronary intervention (PCI). Designed to […]
Abbott Leads Way in First Clinical Trial of Minimally Invasive Clip-Based Repair System for Leaky Tricuspid Heart Valves
ABBOTT PARK, Ill., Aug. 9, 2017 — Abbott today announced that the first patient has been enrolled in a clinical study to evaluate a minimally invasive clip-based repair system for treating people with moderate or severe tricuspid regurgitation (TR), a common condition affecting the right side of the heart. The […]
Spectranetics Announces Fundamental Change, Make-Whole Adjustment Event, Share Exchange Event and Supplemental Indenture for 2.625% Convertible Senior Notes due 2034
COLORADO SPRINGS, Colo., Aug. 10, 2017 (GLOBE NEWSWIRE) — The Spectranetics Corporation (NASDAQ:SPNC) (“the Company”), a developer and manufacturer of single-use medical devices used in minimally invasive procedures within the cardiovascular system, today announced the occurrence of a Fundamental Change, Make-Whole Adjustment Event and Share Exchange Event, and the execution […]