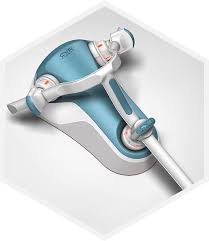
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (NYSE American: CVRS), a leading developer of precision vascular robotics, today reported financial results for the quarter ended June 30, 2017. Recent Highlights and Accomplishments Revenue of $2.3 million in the second quarter 2017 Increased CorPath® GRX installed base to 16 systems with three new […]
Other News
Notice of REVA Medical Briefing Call
SAN DIEGO, Aug. 07, 2017 (GLOBE NEWSWIRE) — REVA Medical, Inc. (ASX:RVA) (“REVA” or the “Company”) will hold a briefing call on Thursday, August 10, 2017 at 4:00 p.m. PDT (which is 9:00 a.m. AEST on Friday, 11 August 2017). Ms. Reggie Groves, the Company’s Chief Executive Officer, will host […]
AUM CARDIOVASCULAR RECEIVES FDA CLEARANCE FOR REVOLUTIONARY DIAGNOSTIC HEART DEVICE
NORTHFIELD, MINNESOTA— AUM Cardiovascular announced that it has received clearance from the Food and Drug Administration for CADence,™ a non-invasive acoustic and ECG device designed to help physicians detect physiological and pathological heart murmurs. The reusable, non-invasive, radiation-free handheld device, which is now available in the United States, records sounds […]
Canon Virginia, Inc. Collaborates with CVR Medical to Manufacture Intuitive, Next-Generation Stroke Prevention Technology
NEWS PROVIDED BY Canon U.S.A., Inc. 10:18 ET NEWPORT NEWS, Va., Aug. 7, 2017 /PRNewswire/ — After recently extending its full range of services to include medical device contract manufacturing and receiving ISO 13485 certification, Canon Virginia, Inc. (CVI), a wholly owned subsidiary of Canon U.S.A., Inc., announces an intent to collaborate with CVR Global […]
Good News For Baseball Pitchers, Other Athletes
CHICAGO, Aug. 7, 2017 /PRNewswire-USNewswire/ — A national study has confirmed the safety of decompression surgery for thoracic outlet syndrome, also known as TOS. It’s good news for patients whose vascular surgeon has recommended surgery. Thoracic outlet syndrome is relatively rare, but is more common among workers and high performance athletes who […]
CeloNova BioSciences Announces Financing Arrangement with Congruent Investment Partners and SWK Holdings Corporation
CeloNova BioSciences Announces Financing Arrangement with Congruent Investment Partners and SWK Holdings Corporation Proceeds to Support Company Growth, Ongoing Clinical Trial Enrollment and New Product Initiatives CeloNova BioSciences, Inc. (CeloNova), a medical device company that develops, manufactures and markets a family of innovative cardiovascular products based upon its novel Polyzene™-F […]
LivaNova (LIVN)’s Perceval Sutureless Aortic Heart Valve Awarded New Technology Add-On Payment From The Center For Medicare And Medicaid Services
LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN) (“LivaNova” or the “Company”), a market-leading medical technology company, today announced its Perceval® sutureless aortic heart valve received approval from the Centers for Medicare and Medicaid Services (CMS) for a New Technology Add-on Payment (NTAP). The Perceval valve met the CMS criteria for NTAP, including the demonstration […]
InspireMD Announces Publication Of An Independent Clinical Review Authored By Leading U.S. And European Physicians Supporting Safety Advantages Of Mesh-Covered Carotid Stents
TEL AVIV, ISRAEL–(Marketwired – August 07, 2017) – InspireMD, Inc. (NYSE American: NSPR) (NYSE MKT: NSPR) (NYSE American: NSPR.WS) (NYSE MKT: NSPR.WS) (“InspireMD” or the “Company”), a leader in embolic prevention systems (EPS) / thrombus management technologies and neurovascular devices, today announced the publication of the independent study entitled, “One swallow does not a […]
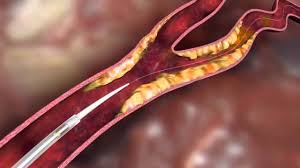
InspireMD’s Cguard Carotid Embolic Prevention System Featured At The SOLACI CACI Congress Of Cardiology 2017 In Buenos Aires
TEL AVIV, ISRAEL–(Marketwired – August 03, 2017) – InspireMD, Inc. (NYSE MKT: NSPR) (NYSE MKT: NSPR.WS) (“InspireMD” or the “Company”), a leader in embolic prevention systems (EPS) / thrombus management technologies and neurovascular devices, today announced that an endovascular interventional procedure featuring the CGuard™ EPS performed by the team of Dr. Anibal Damonte, Interventional […]
RTI Surgical® Divests Cardiothoracic Closure Business to A&E Medical Corporation for up to $60 Million
ALACHUA, Fla.–(BUSINESS WIRE)–RTI Surgical Inc. (RTI) (Nasdaq: RTIX), a global surgical implant company, today announced that it has sold its cardiothoracic closure business to A&E Advanced Closure Systems, LLC (A&E Medical), a leading cardiovascular medical device OEM and a portfolio company of Vance Street Capital LLC, for $54 million in cash […]