MINNEAPOLIS, May 04, 2026 (GLOBE NEWSWIRE) — CVRx, Inc. (NASDAQ: CVRX), a medical device company focused on developing, manufacturing and commercializing innovative neuromodulation solutions for patients with cardiovascular diseases, today announced the enrollment of the first patient in the landmark BENEFIT-HF trial at North Central Heart – a division of the Avera Heart Hospital, in Sioux Falls, S.D. by Dr. Orvar Jonsson. BENEFIT-HF is expected to be one of the largest therapeutic cardiac device trials ever performed in heart failure, enrolling 2,500 patients at approximately 150 centers in the United States and Germany. The prospective, randomized, controlled, multicenter trial is supported by CMS Category B IDE coverage and designed to evaluate all-cause mortality and heart failure decompensation events in a significantly expanded heart failure population. “We are honored to enroll the first patient in the BENEFIT-HF trial,” said Dr. Orvar Jonsson, North Central Heart – a division of the Avera Heart Hospital. “This study will evaluate Barostim therapy in a broader population of patients who remain symptomatic despite optimized guideline-directed medical therapy, generating important data that will further define Barostim’s position in the heart failure continuum.” “Achievement of this early milestone reflects tremendous excitement in the healthcare community regarding this landmark trial, and strong execution by the CVRx team in partnership with BENEFIT-HF investigators,” said Kevin Hykes, President and Chief Executive Officer of CVRx. “We are proud to collaborate with leading physicians in development of such meaningful clinical evidence, and excited to further characterize the patient benefits of Barostim therapy that we believe will drive long-term adoption.” The BENEFIT-HF trial is expected to continue through 2032. If successful, the Trial could expand the indicated patient population for Barostim by approximately three times, significantly broadening access to the therapy. About CVRx, Inc.CVRx is a commercial-stage medical device company focused on developing, manufacturing and commercializing innovative neuromodulation solutions for patients with cardiovascular diseases. Barostim™ is the first medical technology approved by FDA that uses neuromodulation to improve the symptoms of patients with heart failure. Barostim is an implantable device that delivers electrical pulses to baroreceptors located in the wall of the carotid artery. The therapy is designed to restore balance to the autonomic nervous system and thereby reduce the symptoms of heart failure. Barostim received the FDA Breakthrough Device designation and is FDA-approved for use in heart failure patients in the U.S. It has been certified as compliant with the EU Medical Device Regulation (MDR) and holds CE Mark approval for heart failure and resistant hypertension in the European Economic Area. To learn more about Barostim, visit www.cvrx.com. Forward-Looking StatementsThis press release contains forward-looking statements, including statements regarding the expected timing, enrollment, scope, and outcomes of the BENEFIT-HF clinical trial, potential expansion of the Barostim indication, and anticipated benefits of Barostim therapy. These forward-looking statements speak only as of the date of this press release and are subject to a number of known and unknown risks, uncertainties and assumptions, including, but not limited to, our expectations regarding enrollment; the resulting impact on our addressable market; impacts on adoption and regulatory approvals resulting from additional long-term clinical data about our product, including those resulting from the BENEFIT-HF clinical trial; and other important factors that could cause actual results, performance or achievements to differ materially from those that are found in “Part I, Item 1A. Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2025, as such factors may be updated from time to time in our other filings with the Securities and Exchange Commission. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. Investor Contact:Mark Klausner or Mike VallieICR Healthcare443-213-0501ir@cvrx.com Media Contact:Emily MeyersCVRx, Inc.763-416-2853emeyers@cvrx.com
Other News
Cytokinetics to Announce Topline Results from ACACIA-HCM, the Pivotal Phase 3 Clinical Trial of Aficamten in Patients with Non-Obstructive Hypertrophic Cardiomyopathy, on May 5, 2026
Company to Host Conference Call and Webcast Tuesday May 5 at 8:00 AM Eastern Time SOUTH SAN FRANCISCO, Calif., May 04, 2026 (GLOBE NEWSWIRE) — Cytokinetics, Incorporated (Nasdaq: CYTK) today announced it will report topline results from ACACIA-HCM (Assessment Comparing Aficamten to Placebo on Cardiac Endpoints In Adults with Non-Obstructive HCM) on Tuesday, May 5, 2026. Following the announcement, Cytokinetics will host a conference call at 8:00 AM Eastern Time to discuss the results. The conference call will be simultaneously webcast and can be accessed from the Investors & Media section of Cytokinetics’ website at https://ir.cytokinetics.com or the following link: ACACIA-HCM Topline Results. An archived replay of the webcast will be available via Cytokinetics’ website for six months. About Cytokinetics Cytokinetics is a specialty cardiovascular biopharmaceutical company, building on its over 25 years of pioneering scientific innovations in muscle biology, and advancing a pipeline of potential new medicines for patients suffering from diseases of cardiac muscle dysfunction. Cytokinetics’ MYQORZO® (aficamten) is a cardiac myosin inhibitor approved in the U.S., Europe and China for the treatment of adults with symptomatic obstructive hypertrophic cardiomyopathy (oHCM). Cytokinetics is also developing omecamtiv mecarbil, an investigational cardiac myosin activator for the potential treatment of patients with heart failure with severely reduced ejection fraction and ulacamten, an investigational cardiac myosin inhibitor for the potential treatment of heart failure with preserved ejection fraction, while continuing pre-clinical research and development in muscle biology. For additional information about Cytokinetics, visit www.cytokinetics.com and follow us on X, LinkedIn, Facebook and YouTube. Disclaimer Omecamtiv mecarbil and ulacamten are investigational medicines. They have not been approved nor determined to be safe or efficacious for any disease state or any indication by FDA or any other regulatory agency. Forward-Looking Statements This press release contains forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995 (the “Act”). Cytokinetics disclaims any intent or obligation to update these forward-looking statements and claims the protection of the Act’s Safe Harbor for forward-looking statements. Examples of such statements include, but are not limited to, statements relating to Cytokinetics’ and its partners’ research and development activities of Cytokinetics’ product candidates. Such statements are based on management’s current expectations, but actual results may differ materially due to various risks and uncertainties, including, but not limited to the risks related to Cytokinetics’ business outlines in Cytokinetics’ filings with the Securities and Exchange Commission. Forward-looking statements are not guarantees of future performance, and Cytokinetics’ actual results of operations, financial condition and liquidity, and the development of the industry in which it operates, may differ materially from the forward-looking statements contained in this press release. Any forward-looking statements that Cytokinetics makes in this press release speak only as of the date of this press release. Cytokinetics assumes no obligation to update its forward-looking statements whether as a result of new information, future events or otherwise, after the date of this press release. CYTOKINETICS® and the CYTOKINETICS C-shaped logo are registered trademarks of Cytokinetics in the U.S. and certain other countries. MYQORZO® is a registered trademark of Cytokinetics in the U.S. and the European Union. Contact:Cytokinetics Diane WeiserSenior Vice President, Corporate Affairs(415) 290-7757
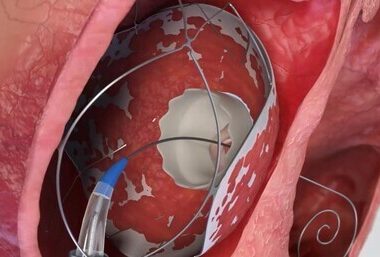
TruLeaf Medical, a fully-owned subsidiary of Allmed Solutions successfully Completed the Second Stage of valve implantation in two Additional Patients
In a follow up of up to nine months, TruLeaf Proprietary RoseDoc Docking System Demonstrates Exceptional Clinical Safety and Efficacy for transcatheter tricuspid valve replacement. OR YEHUDA, Israel, May 4, 2026 /PRNewswire/ — TruLeaf Medical, Ltd, a fully-owned subsidiary of Allmed…
Pulse Biosciences Reports Business Updates and First Quarter 2026 Financial Results
HAYWARD, Calif.–(BUSINESS WIRE)–Pulse Biosciences, Inc. (Nasdaq: PLSE), developer of novel nPulse™ technology using proprietary Nanosecond Pulsed Field Ablation™ (nanosecond PFA or nsPFA™) energy, today announced business updates and financial results for the first quarter ended March 31, 2026. Recent Business Highlights Endocardial Catheter AF Ablation Announced strategic prioritization of the nPulse Cardiac Catheter System following landmark clinical data, presented at AF Symposium, demonst
InspireMD Announces FDA Approval of Investigational Device Exemption Application for CGUARDIANS III Pivotal Study of the SwitchGuard Neuro Protection System
MIAMI, May 01, 2026 (GLOBE NEWSWIRE) — InspireMD, Inc. (Nasdaq: NSPR) (“InspireMD” or the “Company”), developer of the CGuard® Prime carotid stent system for the prevention of stroke, today announced that the U.S. Food and Drug Administration (“FDA”) has approved the company’s Investigational Device Exemption (“IDE”) Application to initiate the CGUARDIANS III pivotal study of its SwitchGuard neuro protection system (“NPS”), including next generation enhancements from our prior IDE approval for use with its CGuard Prime 80 cm stent platform, in transcarotid artery revascularization (“TCAR”) procedures.
Retia Medical Presents First Public Results for Non-Invasive Cardiac Output Algorithm
WHITE PLAINS, N.Y., May 1, 2026 /PRNewswire/ — Retia Medical today announced the first public presentation of results from its non-invasive cardiac output algorithm, which estimates cardiac output using signals from sensors already placed on patients as standard of care: a pulse oximeter…
Echo IQ Announces EchoSolv AS Deployed into Mount Sinai Health System
EchoSolv AS deployed into Mount Sinai Health System, a leading US health system in New YorkMount Sinai Health System comprises seven hospitals, over 400 outpatient practices and +3,760 beds Mount Sinai Fuster Heart Hospital at The Mount Sinai Hospital is ranked No. 1 in New York and No. 2 in the nation for Cardiology, Heart & Vascular Surgery in U.S. News & World Report’s 2025–26 Best Hospitals rankings Mount Sinai Health System has a large echocardiography footprint and top-tier cardiovascular programAgreement represents another milestone in EIQ’s US commercial strategy to deploy in leading hospital systemsEIQ continues to engage with prospective US hospitals regarding potential EchoSolv AS opportunities SYDNEY, April 30, 2026 (GLOBE NEWSWIRE) — AI and Medical Technology company Echo IQ (“the Company”) (ASX: EIQ) is pleased to advise it has deployed its AI-based decision support software for the assessment of severe aortic stenosis, EchoSolv™ AS, with Mount Sinai Health System in New York, USA. “We are proud to work with Mount Sinai Health System, an institution recognised for excellence in cardiovascular care, clinical innovation, and leadership in medicine,” said Nick Lubbers, US President and General Manager for Echo IQ. “This deployment is an important step in our US commercial growth and underscores increasing interest in solutions that can support cardiologists within routine echocardiography workflows. We believe EchoSolv AS offers a practical, measurement-based second look that can help physicians assess severe aortic stenosis more consistently and look forward to working with The Mount Sinai Health System to demonstrate its utility at scale. “Mount Sinai is the kind of institution that helps set the standard for clinical excellence and innovation. For Echo IQ, this engagement represents more than a single large-scale deployment. It reflects our broader growth trajectory as we continue building partnerships with leading hospitals and expanding access to AI-enabled decision support in cardiology,” concluded Lubbers. Stamatios Lerakis MD, PhD*, Director of the Cardiovascular Imaging Program and Imaging for Structural and Valve Interventions for Mount Sinai Health System, said: “We are excited about the opportunity to bring EchoSolv AS into our echocardiography workflow for the evaluation of patients with aortic stenosis. This product can highlight patients with aortic stenosis that can be otherwise under-diagnosed, remain untreated and as a result, not benefit from the tremendous therapeutic advancements in the space of aortic valve diseases. I see this as an adjunct for the echocardiographer who will always have the final say on the interpretation! This product will only strengthen patient care.” The deployment marks a significant milestone for Echo IQ and reflects the Company’s continued growth as health systems and cardiology practices seek practical AI products that can be integrated into existing clinical workflows. Collaboration with a leading medical system such as Mount Sinai Health System also supports the Company’s stated strategy of expanding EchoSolv AS adoption at prominent cardiovascular institutions. Echo IQ views the deployment as an important milestone in not only building commercial traction but also providing future research opportunities as the Company pursues new product development opportunities. The Company continues to engage with prospective US healthcare providers regarding potential EchoSolv AS opportunities and will provide updates as appropriate. Mount Sinai Health System comprises seven hospitals, more than 400 outpatient practices and a globally recognised medical school, the Icahn School of Medicine at Mount Sinai. Specifically, Mount Sinai Fuster Heart Hospital at The Mount Sinai Hospital is ranked No. 1 in New York and No. 2 in the nation for Cardiology, Heart & Vascular Surgery in U.S. News & World Report’s 2025–26 Best Hospitals rankings. The system services millions of patient interactions annually, encompasses over 3,760 beds across campuses, and is consistently ranked among the top hospitals in the US across multiple specialties, including cardiology and heart surgery. Its scale, research pedigree and clinical leadership position it as a high-volume, high-complexity healthcare network at the forefront of adopting advanced digital health and AI-enabled technologies. About EchoSolv AS: EchoSolv AS is an AI-based decision support software indicated for use as an adjunct to echocardiography for the assessment of severe aortic stenosis. When used by an interpreting physician, it provides information to facilitate accurate diagnosis and support clinical review. EchoSolv AS is intended for qualified healthcare professionals and is not intended to replace physician judgment. Authorised for release by the Board of Directors of Echo IQ Limited. Investor Enquiries: Andrew Grover, Executive ChairHenry Jordan, Six Degrees Investor RelationsAndrew.grover@echoiq.ai / investor@echoiq.aiHenry.jordan@sdir.com.au / +61 (0) 431 271 538 ABOUT ECHO IQEcho IQ uses AI-driven technology and proprietary software to improve decision making in Cardiology. The company is based in Sydney, Australia. Disclosures: *Dr. Stamatios Lerakis serves as a paid key opinion leader, speaker and advisory board member for Echo IQ Limited. Forward-Looking Statements: This press release contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995 and for the purposes of ASX Listing Rule 3.1 and ASX Guidance Note 8. Forward-looking statements are statements that are not historical facts and may include, without limitation, statements regarding Echo IQ’s commercial growth strategy, anticipated expansion of EchoSolv AS adoption, development of partnerships with health systems and cardiology practices, expected market adoption of AI-enabled decision support tools, and the potential benefits of EchoSolv AS for clinical decision-making and patient care. These forward-looking statements reflect the current expectations, assumptions, and beliefs of Echo IQ Limited based on information currently available to the company. Forward-looking statements are subject to known and unknown risks, uncertainties, and other factors that may cause actual results, performance, or achievements to differ materially from those expressed or implied by such statements. Such factors include, but are not limited to:Regulatory Risks: the ability of Echo IQ to maintain FDA 510(k) clearance for EchoSolv AS, obtain or maintain any additional required regulatory approvals in the United States or other jurisdictions, and achieve reimbursement coverage from government and private payors;Commercial Risks: the rate at which health systems and cardiology practices adopt EchoSolv AS, the ability of the company to execute on existing commercial agreements including with The Mount Sinai Hospital, performance under and renewal of customer contracts, and the competitive environment for AI-enabled medical software;Financial Risks: the availability of sufficient funding to execute the company’s commercial strategy, the timing and magnitude of revenue generation, and the company’s ability to achieve and sustain profitability; andOperational Risks: the performance and reliability of EchoSolv AS technology in real-world clinical environments, the ability to integrate EchoSolv AS into existing clinical workflows, and broader market acceptance of AI-based decision support tools in cardiology. The assumptions underlying the forward-looking statements in this release are made as of the date of this release and are subject to change without notice. Echo IQ cautions that the foregoing list of risk factors is not exhaustive. Investors and prospective investors should carefully consider these factors, as well as those disclosed in Echo IQ’s periodic filings with the ASX and other public disclosures, before making any investment decision. Echo IQ undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events, or otherwise, except as required by applicable law or regulation, including the ASX Listing Rules and the U.S. securities laws. ™ Registered trademark or trademark of Echo IQ Ltd. or its affiliates. © 2025 Echo IQ Ltd. All rights reserved.
FDA Grants Orchestra BioMed Additional Breakthrough Device Designation for AVIM Therapy
Atrioventricular Interval Modulation (“AVIM”) Therapy Food and Drug Administration (“FDA”) Breakthrough Device Designations span the broader population of patients with uncontrolled hypertension despite medication at increased cardiovascular risk and the specific pacemaker-indicated population being evaluated in the BACKBEAT TrialThe addressable U.S. patient population for AVIM Therapy, based on the indications for use specified in its Breakthrough Device Designations, comprises over 7.7 million adults with hypertension despite medicationBreakthrough Device Designation supports favorable reimbursement pathways, including potential eligibility for New Technology Add-on Payment (“NTAP”) and Transitional Pass-Through (“TPT”) payment, which can facilitate broader, more timely patient access and provider adoption NEW HOPE, Pa., April 30, 2026 (GLOBE NEWSWIRE) — Orchestra BioMed Holdings, Inc. (Nasdaq: OBIO) (“Orchestra BioMed” or the “Company”), a biomedical company accelerating high-impact technologies to patients through strategic partnerships with market-leading global medical device companies, today announced that the FDA has granted a second Breakthrough Device Designation (“BDD”) for AVIM Therapy specific to patients with uncontrolled hypertension despite the use of anti-hypertensive medications, and an indication for a pacemaker. Together, the two BDDs for AVIM Therapy cover indications that encompass both the broader population of patients with uncontrolled hypertension despite medication and increased cardiovascular risk as well as the specific pacemaker-indicated population with uncontrolled hypertension being evaluated in the BACKBEAT Global Pivotal Trial (“BACKBEAT Trial”), which Orchestra BioMed is conducting in collaboration with Medtronic (NYSE: MDT). This additional BDD supports strategic optionality for the clinical, regulatory and commercial reimbursement strategies for AVIM Therapy for both the pacemaker population and potential future expansion populations with uncontrolled hypertension and increased cardiovascular risk. “We are pleased to receive this additional Breakthrough Device Designation from the FDA. We believe it is directly aligned with the patient population being studied in our ongoing BACKBEAT Trial which we are executing in collaboration with Medtronic,” said David Hochman, Chairman and Chief Executive Officer of Orchestra BioMed. “Continued alignment from the FDA, alongside our strategic relationship with Medtronic and accelerated enrollment in the BACKBEAT Trial, puts us in a strong position to advance AVIM Therapy to pivotal trial results.” Orchestra BioMed has a strategic collaboration with Medtronic, the global market leader in cardiac pacing therapies, for development and commercialization of AVIM Therapy for the treatment of uncontrolled hypertension in pacemaker-indicated patients. Under the terms of the existing collaboration agreement, Medtronic holds the right of first negotiation to expand its licensing agreement with Orchestra BioMed to obtain global rights to commercialize AVIM Therapy for the treatment of uncontrolled hypertension in patients that do not have an indication for a pacemaker. The FDA Breakthrough Devices Program, which reflects the FDA’s commitment to device innovation and protecting public health, is designed to expedite the development of and provide priority review for innovative medical technologies that have the potential to significantly improve outcomes for patients with serious or life-threatening conditions. To be eligible for this designation, a device must demonstrate the potential to provide more effective treatment or diagnosis of a life-threatening or irreversibly debilitating condition. In addition, the device must meet at least one of the following criteria: it must represent breakthrough technology, have no approved or cleared alternatives, offer significant advantages over existing options, or be determined by the FDA to be in the best interest of patients. Beyond regulatory acceleration, the BDD may also support favorable reimbursement pathways, including eligibility for incremental inpatient reimbursement through the New Technology Add-on Payment and outpatient Transitional Pass-Through payments under the Centers for Medicare & Medicaid Services programs. These mechanisms may help facilitate more timely access to breakthrough technologies while supporting provider adoption and patient access. About Orchestra BioMed Orchestra BioMed is a biomedical innovation company accelerating high-impact technologies to patients through strategic collaborations with market-leading global medical device companies. The Company’s two flagship product candidates – Atrioventricular Interval Modulation (AVIM) Therapy and Virtue® Sirolimus AngioInfusion™ Balloon (Virtue SAB) – are currently undergoing pivotal clinical trials for their lead indications, each representing multi-billion-dollar annual global market opportunities. AVIM Therapy is a bioelectronic treatment for hypertension, the leading risk factor for death worldwide, and is designed to be delivered by a pacemaker and achieve immediate, substantial and sustained reductions in blood pressure in patients with hypertensive heart disease. The Company has a strategic collaboration with Medtronic, one of the largest medical device companies in the world and the global leader in cardiac pacing therapies, for the development and commercialization of AVIM Therapy for the treatment of uncontrolled hypertension in pacemaker-indicated patients. AVIM Therapy has FDA Breakthrough Device Designations for these patients, as well as an estimated 7.7 million total patients in the U.S. with uncontrolled hypertension despite medical therapy and increased cardiovascular risk. Virtue SAB is a highly differentiated, first-of-its-kind non-coated drug delivery angioplasty balloon system designed to deliver a large liquid dose of proprietary extended-release formulation of sirolimus, SirolimusEFR™, for the treatment of atherosclerotic artery disease, the leading cause of mortality worldwide. Virtue SAB has been granted Breakthrough Device Designation by the FDA for the treatment of coronary in-stent restenosis, coronary small vessel disease and below-the-knee peripheral artery disease. For further information about Orchestra BioMed, please visit www.orchestrabiomed.com, and follow us on LinkedIn. About AVIM TherapyAVIM Therapy is an investigational therapy compatible with standard dual-chamber pacemakers designed to substantially and persistently lower blood pressure. It has been evaluated in pilot studies in patients with hypertension who are also indicated for a pacemaker. MODERATO II, a double-blind, randomized pilot study, showed that patients treated with AVIM Therapy experienced net reductions of 8.1 mmHg in 24-hour ambulatory systolic blood pressure (aSBP) and 12.3 mmHg in office systolic blood pressure (oSBP) at six months when compared to control patients. In addition to reducing blood pressure, clinical results using AVIM Therapy demonstrate improvements in cardiac function and hemodynamics. The BACKBEAT (BradycArdia paCemaKer with atrioventricular interval modulation for Blood prEssure treAtmenT) global pivotal study will evaluate the safety and efficacy of AVIM Therapy in lowering blood pressure in patients who have systolic blood pressure above target despite anti-hypertensive medication and who are indicated for or have recently received a dual-chamber cardiac pacemaker. AVIM Therapy has been granted two Breakthrough Device Designations by the FDA for the treatment of uncontrolled hypertension in patients who have increased cardiovascular risk. Forward-Looking Statements Certain statements included in this press release that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995. Forward-looking statements generally are accompanied by words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “seem,” “seek,” “future,” “outlook” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements relating to the initiation, enrollment, timing, implementation and design of the Company’s ongoing pivotal trials, realizing the clinical and commercial value of AVIM Therapy and Virtue SAB, the potential safety and efficacy of the Company’s product candidates, the potential benefits of BDD, including its ability to expedite FDA reviews and support favorable reimbursement pathways, and the ability of the Company’s partnerships to accelerate clinical development. These statements are based on various assumptions, whether or not identified in this press release, and on the current expectations of the Company’s management and are not predictions of actual performance. These forward-looking statements are provided for illustrative purposes only and are not intended to serve as and must not be relied on as a guarantee, an assurance, a prediction, or a definitive statement of fact or probability. Actual events and circumstances are difficult or impossible to predict and may differ from assumptions. Many actual events and circumstances are beyond the control of the Company. These forward-looking statements are subject to a number of risks and uncertainties, including changes in domestic and foreign business, market, financial, political, and legal conditions; risks related to regulatory approval of the Company’s commercial product candidates and ongoing regulation of the Company’s product candidates, if approved; the timing of, and the Company’s ability to achieve expected regulatory and business milestones; the impact of competitive products and product candidates; and the risk factors discussed under the heading “Item 1A. Risk Factors” in the Company’s Annual Report on Form 10-K for the year ended December 31, 2025, which was filed with the SEC on March 12, 2026. The Company operates in a very competitive and rapidly changing environment. New risks emerge from time to time. Given these risks and uncertainties, the Company cautions against placing undue reliance on these forward-looking statements, which only speak as of the date of this press release. The Company does not plan and undertakes no obligation to update any of the forward-looking statements made herein, except as required by law. Investor Contact:Silas NewcombOrchestra BioMedSnewcomb@orchestrabiomed.com Media Contact:Kelsey KirkOrchestra BioMedkkirkellis@orchestrabiomed.com
New AccuCode CV Platform Brings Specialty-Specific Intelligence to Cardiovascular Coding
JACKSONVILLE BEACH, Fla.–(BUSINESS WIRE)–MedAxiom and AccuCode AI today announced the launch of AccuCode CV, an artificial intelligence (AI)-augmented cardiovascular coding engine that optimizes revenue integrity by improving accuracy, decreasing turnaround times and reducing costs. This first-of-its-kind platform applies specialty-specific intelligence to the most complex and highest-risk cardiovascular coding scenarios, helping provider organizations improve coding accuracy, efficiency, qua
VVT Med Announces Positive Clinical Data for ScleroSafe(R) Published in Peer-Reviewed Indian Journal of Vascular and Endovascular Surgery
Prospective Study Conducted at Holy Spirit Hospital in Mumbai Reports 95% Vein Occlusion Rate at 30 Prospective Study Conducted at Holy Spirit Hospital in Mumbai Reports 95% Vein Occlusion Rate at 30