Apr 29, 2026 First-in-Class enVVe system to be evaluated in TAVVE U.S. Pivotal Trial Clinical site activation and patient enrollment expected to commence later this year Large unmet clinical need for approximately 3 million U.S. patients with severe deep Chronic Venous Insufficiency Potential multi-billion-dollar untapped U.S. market Company has approximately […]
Other News
Rad AI Appoints Chief Operating Officer and First Chief Clinical Officer, Strengthening Operational and Clinical Strategy
The appointments signal Rad AI’s commitment to operational excellence and physician-led clinical innovation SAN FRANCISCO, April 29, 2026 /PRNewswire/ — Rad AI, the leader in AI-powered radiology workflow solutions, today announced David Leonard as the company’s Chief Operating Officer…
Adagio Medical to Participate in the BofA Securities 2026 Health Care Conference
LAGUNA HILLS, Calif.–(BUSINESS WIRE)—- $ADGM #ARRHYTHMIA–Adagio Medical Holdings, Inc. (Nasdaq: ADGM) (“Adagio” or “the Company”), a leading innovator in catheter ablation technologies for the treatment of cardiac arrhythmias, today announced that management will be presenting at the BofA Securities 2026 Healthcare Conference on Wednesday, May 13, 2026 at 4:35pm PDT in Las Vegas. Presentation Information A live webcast and replay of the session will be accessible at https://bofa.veracast.com/webcasts/bofa/health
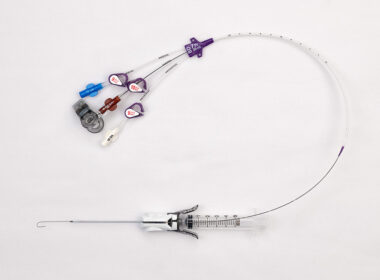
BD Reimagines Central Line Insertion, Advancing Leadership in Patient Safety Through Breakthrough Vascular Access Technology
With the launch of CentroVena One™, BD brings a first‑of‑its‑kind insertion system to market that simplifies central line placement for clinicians and enhances care in acute settings FRANKLIN LAKES, N.J., April 29, 2026 /PRNewswire/ — BD (Becton, Dickinson and Company) (NYSE: BDX), a…
Kestra Medical Technologies to Present at the BofA Securities 2026 Healthcare Conference
KIRKLAND, Wash., April 29, 2026 (GLOBE NEWSWIRE) — Kestra Medical Technologies, Ltd. (Nasdaq: KMTS), a leading wearable medical device and digital healthcare company, is scheduled to present at the BofA Securities 2026 Healthcare Conference on Wednesday, May 13 at 2:20 p.m. Pacific Time. Interested parties may access the live webcast and a replay of the presentation by visiting the Kestra investor relations website. About Kestra Medical TechnologiesKestra Medical Technologies is a leading wearable medical device and digital healthcare company focused on transforming patient outcomes in cardiovascular disease using monitoring and therapeutic intervention technologies that are intuitive, intelligent, and connected. For more information, visit www.kestramedical.com. CONTACT: Investor contact
Neil Bhalodkar
neil.bhalodkar@kestramedical.com
Boehringer Ingelheim and Eko Health Inc. launch new AI-based solution that detects heart murmurs in dogs
Eko Vet+TM | CANINEBEAT® AI helps veterinarians detect, visualize, and grade heart murmurs in dogs that can otherwise go undetectedThe CANINEBEAT® AI algorithm detects heart murmurs associated with structural heart disease in dogs with more than 95% sensitivity and specificity*With earlier murmur detection, dogs can benefit from heart disease diagnostics and treatment sooner INGELHEIM, Germany, and SAN FRANCISCO, US – Boehringer Ingelheim, a global leader in animal health, and Eko Health, a leader in AI-powered cardiac and pulmonary disease detection, announced the launch of an innovative solution to detect, visualize, and grade heart murmurs in dogs. Eko Vet+TM | CANINEBEAT® AI aims to improve health outcomes for dogs with heart disease by enabling veterinarians to identify heart murmurs that might otherwise go unnoticed. “Our proprietary CANINEBEAT® AI algorithm integrated into the Eko Vet+TM app will powerfully support veterinarians’ early detection of heart murmurs in dogs and pet owner conversations,” said Dr. Erich Schött, Head of the Pet Business at Boehringer Ingelheim. “Heart murmur detection in dogs can be quite difficult, especially in busy clinics. Because pet owners don’t see clinical signs in the early stage of heart disease, it’s a challenge for veterinarians to explain that the dog suffers from a serious disease. With this unique AI solution, we can help detect murmurs earlier in the disease process, so dogs may receive appropriate diagnosis and treatment sooner.” The new digital solution includes three integrated components: ● The Eko CORE™ Digital Attachment connects to most single-tube stethoscopes and digitizes heart sounds that can then be analyzed by AI-based algorithms. It also helps veterinarians hear more subtle murmurs by amplifying heart sounds by 40 times and offers advanced noise cancellation and cardiac sound filtering. Other Eko devices also are compatible with the veterinary digital solution. ● The CANINEBEAT® AI algorithm, trained and validated on more than 4,000 annotated canine heart sound recordings, detects heart murmurs associated with structural heart disease in dogs with more than 95% sensitivity and specificity.* The algorithm is intended to support veterinary clinical assessment and does not replace comprehensive cardiac evaluation or professional veterinary judgment.● The EkoVet+TM app incorporates insights from the CANINEBEAT® AI algorithm, including murmur images, sound files, and shareable reports. The app provides clinical support and helps veterinarians discuss the potential findings and any diagnostics and treatment needs with pet owners. Heart disease affects approximately 10% of all dogs.1 The most common heart disease in dogs is myxomatous mitral valve disease (MMVD), which often produces a heart murmur when the heart has a leak in one of the heart valves, preventing blood from getting pumped correctly. MMVD is a chronic condition that is a major cause of morbidity and mortality in dogs.2 Because dogs in early stages of heart disease may be asymptomatic, heart murmurs can go unnoticed during routine examinations. To address this challenge, 50 global veterinary cardiology experts contributed to the development of Eko Vet+TM with CANINEBEAT® AI. “CANINEBEAT® AI inside Eko Vet+TM is an exceptional resource for general practitioner veterinarians, helping them quickly detect and grade murmurs with a high level of consistency,” said Prof. Gerhard Wess, Head of the Cardiology Service, Clinic for Small Animal Internal Medicine, Ludwig-Maximilians-Universität in Munich. “I am thrilled to be involved in the development of this new AI-powered solution that will give veterinarians greater confidence in heart murmur detection and support better outcomes for dogs.” The introduction of this innovative auscultation solution for the veterinary industry has begun in the United States and the United Kingdom and will start in Germany next month. A phased expansion is planned in additional markets later this year and next. It will be available through both Boehringer Ingelheim and Eko Health, comprising the same three components through distinct commercial offers. “We’ve seen firsthand in human health how earlier, more accurate detection of cardiac disease can change the trajectory of care,” said Connor Landgraf, CEO and co-founder of Eko Health. “Collaborating with Boehringer Ingelheim, a leader in canine cardiology, allows us to bring that same innovative approach to veterinary medicine. Together, we’re equipping veterinarians with AI-powered tools to identify heart murmurs earlier and support better outcomes in canine patients.” About Boehringer Ingelheim – Animal Health business Boehringer Ingelheim provides innovation for preventing and treating diseases in animals. The company offers a wide range of vaccines, parasite-control products, and medicines for pets, horses, and livestock to veterinarians, animal owners, farmers, and governments. As a leader in animal health, Boehringer Ingelheim values that the health of humans and animals is deeply connected and strives to make a difference for people, animals, and society. Learn more at www.boehringer-ingelheim.com/animal-health. About Boehringer IngelheimBoehringer Ingelheim is a biopharmaceutical company active in both human and animal health. As one of the industry’s top investors in research and development, the company focuses on developing innovative therapies that can improve and extend lives in areas of high unmet medical need. Independent since its foundation in 1885, Boehringer takes a long-term perspective, embedding sustainability along the entire value chain. Our approximately 54,300 employees serve over 130 markets to build a healthier and more sustainable tomorrow. Learn more at www.boehringer-ingelheim.com. About Eko HealthEko Health is a pioneering AI early disease detection company advancing how healthcare professionals detect and monitor heart and lung disease with its portfolio of digital stethoscopes, ECG devices, software, and AI-powered analysis. Its FDA-cleared platform, with more than 700,000 devices sold worldwide, helps clinicians detect earlier and manage treatment more effectively to improve patient outcomes. Eko Health is headquartered in Emeryville, California. For more information, visit www.ekohealth.com. Media Contacts:Mi-Kyung Lee Langemi-kyung.lee-lange@boehringer-ingelheim.com Sam Moore sam.moore@ekohealth.com Editorial Note: * The CANINEBEAT study was conducted using one of the largest canine digital auscultation datasets to date, comprising more than 4,000 heart sound recordings collected from over 3,400 dogs. The study assessed the performance of the CANINEBEAT® AI algorithm in detecting canine heart murmurs. All recordings were systematically annotated, with the algorithm’s development and validation supported by nearly 50 veterinary cardiology experts. Detailed study results will be presented and published in peer reviewed scientific forums. References: 1 Keene, Bruce W et al. “ACVIM consensus guidelines for the diagnosis and treatment of myxomatous mitral valve disease in dogs.” Journal of veterinary internal medicine vol. 33,3 (2019): 1127-1140. https://doi.org/10.1111/jvim.154882 Häggström, J et al. “New insights into degenerative mitral valve disease in dogs.” Veterinary Clinics of North America: Small Animal Practice. 2004 vol 34, 5: 1209-1226. doi: 10.1016/J.CVSM.2004.05.002.
Saving Limbs Where Others Stop: Cagent Vascular Announces Landmark Peer-Reviewed Data Demonstrating Safety and Efficacy of Serration Remodeling Therapy™ in the Most Challenging Arteries Below-the-Ankle
WAYNE, Pa.–(BUSINESS WIRE)– #CLI–Cagent Vascular, a leader in innovative endovascular technologies, today announced the publication of a peer-reviewed study in the Journal of Endovascular Therapy (JEVT) demonstrating the safety and efficacy of the Serranator® PTA Serration Balloon Catheter in pedal (inframalleolar) interventions for patients with chronic limb-threatening ischemia (CLTI). The investigator-initiated, multi-institutional, retrospective study, led by Dr. Edward Gifford, vascular surgeo
Cordis Launches SELUTION SLR™ PTA Drug-Eluting Balloon (DEB) in Japan
TOKYO–(BUSINESS WIRE)– #Cordis–Cordis, a global leader in interventional cardiovascular technologies, today announces the launch and availability of the SELUTION SLR™ PTA Drug-Eluting Balloon (DEB)1 for use in Japan. SELUTION SLR™ DEB is designed to deliver sirolimus in a controlled, sustained manner to reduce restenosis and restore vessel function. The SELUTION SFA Japan clinical trial results demonstrate durable clinical outcomes in a complex patient population, including 81.5% primary patency and 9
CorVista Health Announces Collaboration with Mayo Clinic to Advance Next-Gen Pulmonary Hypertension Diagnosis
BETHESDA, Md.–(BUSINESS WIRE)–CorVista Health has announced a research collaboration with Mayo Clinic to evaluate how next-generation, non-invasive diagnostics may transform the identification and management of pulmonary hypertension (PH) – a complex and frequently underdiagnosed cardiovascular condition that affects an estimated one percent of the global population. Pulmonary hypertension is a progressive and potentially life-threatening condition characterized by elevated pressure in the pu
BridgeBio Launches Health Education Effort on Often Overlooked Heart Condition Featuring Attruby® Advocates, Morgan Freeman and Sports Business Icon Howard H. White
– New national campaign encourages recognition of ATTR-CM, often mistaken for other heart conditions, where earlier diagnosis and treatment may help improve outcomes1 PALO ALTO, Calif., April 28, 2026 (GLOBE NEWSWIRE) — BridgeBio Pharma, Inc. (Nasdaq: BBIO) (the “Company,” “we” or “BridgeBio”), a biopharmaceutical company focused on developing medicines for genetic conditions, has teamed up with award-winning actor Morgan Freeman and Howard “H” White, a longtime basketball apparel marketing executive, cultural connector, and heart health advocate, to bring greater attention to ATTR-CM, a progressive heart disease that can be underrecognized and often mistaken for other forms of heart failure.1,2 Don’t Pass On Your Heart Health encourages people to take persistent symptoms seriously, recognizing that earlier action can help support better long-term outcomes. Although ATTR-CM has historically been diagnosed more often in men, research suggests the need for broader awareness across men and women over 55.3 “In clinical practice, ATTR-CM is often hiding in plain sight. Many people come in with symptoms like shortness of breath, fatigue, carpal tunnel, nerve pain, or leg swelling that are commonly attributed to other forms of heart failure or aging,” said Albert J. Hicks, III, M.D., MPH, Section Chief of Heart Failure & Cardiac Transplant at University of Maryland Medical Center. “What’s critical is recognizing when those symptoms may point to an underlying condition that requires further evaluation. Increasing awareness among both clinicians and patients can help prompt earlier recognition, which is an important step toward improving long-term outcomes.” “We continue to see a disproportionate impact of ATTR-CM in Black communities, where people may face delays in diagnosis and access to care,” said Richard Allen Williams, M.D., FACC, Founder and past President of the Association of Black Cardiologists. “Improving awareness is critical, but so is ensuring people feel empowered to speak up, ask questions, and advocate for themselves when something doesn’t feel right.” Howard “H” White is known for shaping athlete-driven movements in global sports culture. Over a four-decade marketing career, he is an author, philanthropist and is widely recognized for building connections between sports, community, and culture, including founding youth-focused initiatives that empower the next generation. Howard experienced symptoms not typically associated with heart failure or heart disease, including carpal tunnel syndrome and an enlarged heart. As his condition progressed, he developed shortness of breath, leg swelling, and fatigue, and by the time he recognized the seriousness of his symptoms, his disease had significantly advanced. He is now committed to ensuring others do not go through the same experience, encouraging anyone experiencing these symptoms to speak with their doctor early so it does not progress to that point. His commitment to advancing awareness and care also includes founding the Howard “H” White Center for Cardiac Amyloidosis at Providence St. Vincent Medical Center, which focuses on improving diagnosis, treatment, and education around amyloid heart disease. “For years, people have been living with cardiac amyloidosis without knowing it,” said Howard “H” White. “I didn’t recognize it until simple things – like walking up the stairs – left me out of breath. I had been told my enlarged heart was from sports. The truth was, amyloid was making my heart bigger and weaker. By the time I found out, my condition had already progressed significantly. Today, earlier detection is possible. My message is simple: know the signs, ask questions, and see your doctor so you don’t have to follow my path.” Transthyretin (TTR) is a protein that circulates in the blood, where it transports thyroxine and vitamin A and supports critical functions, including cognition, cardiovascular health, muscle integrity, metabolism, vision, and bone strength. ATTR-CM occurs when misfolded TTR protein, also known as amyloid, builds up in the heart over time. As deposits accumulate, the heart can become thicker and stiffer, making it harder to pump blood effectively. Because symptoms may resemble those of more common forms of heart failure, or be dismissed as “normal aging,” ATTR-CM is frequently overlooked or identified later in the disease journey.3 ATTR-CM includes both wild-type disease associated with aging and hereditary (variant) forms caused by genetic changes in the transthyretin (TTR) gene.1 Certain variants are more commonly carried by people of African ancestry, and approximately 1 in 25 Black/African American individuals in the United States carry the V142I (also known as V122I) variant associated with ATTR-CM.2 As a result, Black men and women over 55 may face elevated risk and delayed diagnosis, with research showing that people with this variant can experience more severe disease and poorer outcomes, including worse survival and higher rates of heart failure.2,4,5 Recent clinical findings highlight why recognizing ATTR-CM earlier can make a meaningful difference. In the Phase 3 ATTRibute-CM study evaluating Attruby®, the treatment showed consistent benefit in people with both wild-type and hereditary forms of the disease.6,7 Changes linked to stabilizing the TTR protein were seen as early as four weeks after starting treatment and were sustained over time.8 Over the course of the study, Attruby helped adults with ATTR-CM live longer and have fewer hospitalizations due to heart issues. 9 At 30 months, treatment was associated with a 50% reduction in the cumulative frequency of cardiovascular-related hospitalizations compared to placebo, and people taking Attruby also reported a better health-related quality of life compared to those not taking Attruby.6,7,10 The majority of side effects, like diarrhea and abdominal pain, were mild and went away on its own, allowing people to stay on Attruby.9 “ATTR-CM is a condition where earlier awareness can make a meaningful difference, yet many people are still diagnosed only after symptoms have progressed,” said Jonathan Fox, M.D., Ph.D., Chief Medical Officer at BridgeBio Cardiovascular. “This is particularly important for individuals carrying genetic variants, such as p.Val142Ile (also known as V122I), who often experience delays in diagnosis and limited awareness, which can contribute to poorer outcomes over time. Improving recognition of the disease, especially at a stage when intervention may help stabilize disease progression, is an important step toward better long-term outcomes.” To support people in taking the next step for their heart health, BridgeBio has launched a new educational hub on Attruby.com offering clear information about ATTR-CM including Howard “H” White’s testimonial video, practical resources, and a downloadable Doctor Discussion Guide to help individuals and families recognize potential warning signs and prepare for informed conversations with healthcare providers. BridgeBio continues research to better understand outcomes in both wild-type and variant ATTR-CM, helping to advance knowledge of how the disease affects different populations. To hear more from Howard “H” White on the importance of recognizing ATTR-CM earlier, visit: attruby.com/howard-h-white About Attruby® (acoramidis) INDICATIONAttruby is a transthyretin stabilizer indicated for the treatment of the cardiomyopathy of wild-type or variant transthyretin-mediated amyloidosis (ATTR-CM) in adults to reduce cardiovascular death and cardiovascular-related hospitalization.7 IMPORTANT SAFETY INFORMATION Adverse ReactionsDiarrhea (11.6% vs 7.6%) and upper abdominal pain (5.5% vs 1.4%) were reported in patients treated with Attruby versus placebo, respectively. The majority of these adverse reactions were mild and resolved without drug discontinuation. Discontinuation rates due to adverse events were similar between patients treated with Attruby versus placebo (9.3% and 8.5%, respectively).7 About BridgeBio Pharma, Inc.BridgeBio exists to develop transformative medicines for genetic conditions. Millions of people worldwide living with genetic conditions lack treatment options, often because drug development for small patient populations can be commercially challenging. We aim to bridge the gap between advancements in genetic science and meaningful medicines for underserved patient populations. Our decentralized, hub-and-spoke model is designed for speed, precision, and scalability. Autonomous and empowered teams focus on individual conditions, while a central hub provides the clinical, regulatory, and commercial capabilities needed to bring innovation to market. For more information, visit bridgebio.com and follow us on LinkedIn, X, Facebook, Instagram, YouTube, and TikTok. BridgeBio Pharma, Inc. Forward-Looking StatementsThis press release contains forward-looking statements. Statements in this press release may include statements that are not historical facts and are considered forward-looking within the meaning of Section 27A of the Securities Act of 1933, as amended (the Securities Act), and Section 21E of the Securities Exchange Act of 1934, as amended (the Exchange Act), which are usually identified by the use of words such as “anticipates,” “believes,” “continues,” “estimates,” “expects,” “hopes,” “intends,” “may,” “plans,” “projects,” “remains,” “seeks,” “should,” “will,” and variations of such words or similar expressions. BridgeBio intends these forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act and Section 21E of the Exchange Act. These forward-looking statements include statements regarding the potential benefits of earlier awareness, recognition, diagnosis and treatment of ATTR-CM, the potential ability of intervention to help stabilize disease progression or improve long-term outcomes, and the Company’s ongoing and future research regarding outcomes in wild-type and variant ATTR-CM. Although the Company believes that its plans, intentions, expectations and strategies as reflected in or suggested by those forward-looking statements are reasonable, the Company can give no assurance that the plans, intentions, expectations or strategies will be attained or achieved. Furthermore, actual results may differ materially from those described in the forward-looking statements and will be affected by a number of risks, uncertainties and assumptions, including, but not limited to those risks set forth in the Risk Factors section of the Company’s most recent Annual Report on Form 10-K and the Company’s other filings with the U.S. Securities and Exchange Commission. Moreover, the Company operates in a very competitive and rapidly changing environment in which new risks emerge from time to time. These forward-looking statements are based upon the current expectations and beliefs of the Company’s management as of the date of this press release, and are subject to certain risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Except as required by applicable law, BridgeBio assumes no obligation to update publicly any forward-looking statements, whether as a result of new information, future events or otherwise. BridgeBio Contact:Bubba Murarka, Executive Vice Presidentcontact@bridgebio.com (650)789-8220 References: Attruby.com, “What Is ATTR-CM?” Disease overview and symptom information. Accessed January 27, 2026.Attruby.com, “Signs & Symptoms” section. Accessed January 27, 2026.Buxbaum JN, Ruberg FL, Tripp KP, et al. The amyloidogenic V122I transthyretin variant in elderly Black Americans. N Engl J Med. 2014;370(23):2135–2142. Accessed January 27, 2026.Khedraki R, et al. Race, genotype, and prognosis in transthyretin amyloid cardiomyopathy. Am J Cardiol. 2024;216:97–104. doi:10.1016/j.amjcard.2024.02.019. Accessed March 24, 2026.Damrauer SM, Chaudhary K, Cho JH, et al. Association of the TTR V122I Variant With Heart Failure Among Individuals of African or Hispanic/Latino Ancestry. JAMA. 2019;322(22):2191–2202. doi:10.1001/jama.2019.17935. Accessed March 24, 2026Gillmore JD, Judge DP, Cappelli F, et al. Efficacy and safety of acoramidis in transthyretin amyloid cardiomyopathy. N Engl J Med. 2024;390(2):132-142.doi:10.1056/NEJMoa2305434.Attruby® (acoramidis) Prescribing Information. BridgeBio Pharma, Inc. Accessed January 27, 2026.Maurer M, Sarswat N, Grogan M, et al. Acoramidis improves serum TTR levels in patients with wild-type or variant transthyretin amyloid cardiomyopathy–results from ATTRibute-CM. Poster presented at: Annual Congress of the Heart Failure Association of the European Society of Cardiology; May 17-20, 2025; Belgrade, Serbia. Accessed March 24, 2026.BridgeBio Pharma, Inc. (2024). Acoramidis Significantly Reduces All-cause Mortality in the Overall ATTR-CM Variant and V142I (V122I) Populations press release. Accessed February 3, 2026.BridgeBio Pharma, Inc. (2025). Acoramidis Demonstrates Statistically Significant Reduction in Cardiovascular Mortality (CVM) Through Month 42 of the ATTRibute-CM Open-Label Extension. August 30, 2025. Available at: investor.bridgebio.com. Accessed March 30, 2026.