PRINCETON, N.J., June 27, 2024 (GLOBE NEWSWIRE) — Acasti Pharma Inc. (Nasdaq: ACST) (Acasti or the Company), a late-stage, biopharma company advancing GTX-104, its novel injectable formulation of nimodipine that addresses high unmet medical needs for a rare disease, aneurysmal subarachnoid hemorrhage (aSAH), today announced that the Company’s pivotal Phase 3 STRIVE-ON safety trial (the STRIVE-ON trial–NCT05995405) has exceeded the 50% enrollment milestone. The STRIVE-ON trial, a prospective, open-label, randomized (1:1 ratio), parallel group trial of GTX-104 compared with oral nimodipine in 100 patients hospitalized for aSAH, initiated patient enrollment in October of 2023. The primary endpoint is safety and will be measured as comparative adverse events, including hypotension, between the two groups. “Since dosing the first patient in STRIVE-ON last October, we have continued to build momentum by activating high volume neurocritical care hospitals across the country with unrelenting focus on executing patient enrollment and investigator engagement,” said Prashant Kohli, CEO of Acasti. “Achievement of our 50% enrollment milestone reflects laser sharp focus from both our participating clinical trial sites and the Acasti team. Investigators continue to be enthusiastic about the potential of GTX-104 as an IV alternative to oral nimodipine for the treatment of aSAH. Based on a comprehensive review of enrollment factors, we currently anticipate randomizing all 100 patients in late 2024 to early 2025, while staying on track for a potential NDA submission to the FDA in the first half of calendar 2025.” “Patients with aSAH require intensive management and present with a variety of complications that make consistent administration of oral nimodipine difficult especially in patients with severe neurological deficits with dysphasia or requiring mechanical ventilation,” said Dr. Abhishek Ray, Associate Professor of Neurological Surgery at University Hospitals Cleveland, Case Western Reserve University School of Medicine. “GTX-104 shows great promise as an IV alternative to the current standard of care, and we look forward to assessing the data obtained from this trial.” About aneurysmal Subarachnoid Hemorrhage (aSAH) aSAH is bleeding over the surface of the brain in the subarachnoid space between the brain and the skull, which contains blood vessels that supply the brain. A primary cause of such bleeding is the rupture of an aneurysm. Approximately 70% of aSAH patients experience death or dependence, and more than 30% die within one month of hemorrhage. Approximately 50,000 patients in the United States are affected by aSAH per year, based on market research. Outside of the United States, annual cases of aSAH are estimated at approximately 60,000 in the European Union, and approximately 150,000 in China. About the GTX-104 GTX-104 is a clinical stage, novel, injectable formulation of nimodipine being developed for intravenous (IV) infusion in aSAH patients to address significant unmet medical needs. The unique nanoparticle technology of GTX-104 facilitates aqueous formulation of insoluble nimodipine for a standard peripheral IV infusion. GTX-104 provides a convenient IV delivery of nimodipine in the Intensive Care Unit potentially eliminating the need for nasogastric tube administration in unconscious or dysphagic patients. Intravenous delivery of GTX-104 also has the potential to lower food effects, drug-to-drug interactions, and eliminate potential dosing errors. Further, GTX-104 has the potential to better manage hypotension in aSAH patients. GTX-104 has been administered in over 150 healthy volunteers and was well tolerated with significantly lower inter- and intra-subject pharmacokinetic variability compared to oral nimodipine. The addressable market in the United States for GTX-104 is estimated to be about $300 million, based on market research. About Acasti Acasti is a late-stage biopharma company with drug candidates addressing rare and orphan diseases. Acasti’s novel drug delivery technologies have the potential to improve the performance of currently marketed drugs by achieving faster onset of action, enhanced efficacy, reduced side effects, and more convenient drug delivery. Acasti’s lead clinical assets have each been granted Orphan Drug Designation by the FDA, which provides seven years of marketing exclusivity post-launch in the United States, and additional intellectual property protection with over 40 granted and pending patents. Acasti’s lead clinical asset, GTX-104, is an intravenous infusion targeting aneurysmal Subarachnoid Hemorrhage (aSAH), a rare and life-threatening medical emergency in which bleeding occurs over the surface of the brain in the subarachnoid space between the brain and skull. For more information, please visit: www.acasti.com. Forward-Looking Statements Statements in this press release that are not statements of historical or current fact constitute “forward-looking statements” within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, as amended, Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, and “forward-looking information” within the meaning of Canadian securities laws (collectively, “forward-looking statements”). Such forward looking statements involve known and unknown risks, uncertainties, and other factors that could cause the actual results of Acasti to be materially different from historical results or from any future results expressed or implied by such forward-looking statements. In addition to statements which explicitly describe such risks and uncertainties, readers are urged to consider statements containing the terms “believes,” “belief,” “expects,” “intends,” “anticipates,” “estimates”, “potential,” “should,” “may,” “will,” “plans,” “continue”, “targeted” or other similar expressions to be uncertain and forward-looking. Readers are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this press release. The forward-looking statements in this press release, including statements regarding the Company’s anticipated enrollment and NDA submission schedule for the STRIVE-ON trial, GTX-104’s commercial prospects, and GTX-104’s potential to bring enhanced treatment options to patients suffering from aSAH are based upon Acasti’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, including, without limitation: (i) the success and timing of regulatory submissions of the Phase 3 safety trial for GTX-104; (ii) regulatory requirements or developments and the outcome and timing of the proposed NDA application for GTX-104; (iii) changes to clinical trial designs and regulatory pathways; (iv) legislative, regulatory, political and economic developments; and (v) actual costs associated with Acasti’s clinical trials as compared to management’s current expectations. The foregoing list of important factors that could cause actual events to differ from expectations should not be construed as exhaustive and should be read in conjunction with statements that are included herein and elsewhere, including the risk factors detailed in documents that have been and are filed by Acasti from time to time with the Securities and Exchange Commission and Canadian securities regulators. All forward-looking statements contained in this press release speak only as of the date on which they were made. Acasti undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as required by applicable securities laws. For more information, please contact: Acasti Contact: Prashant KohliChief Executive OfficerTel: 450-686-4555Email: info@acastipharma.com www.acasti.com Investor Relations: LifeSci AdvisorsMike MoyerManaging DirectorPhone: 617-308-4306Email: mmoyer@lifesciadvisors.com
Other News
St. Joseph’s Hospital’s ‘Tampa 2’ AFib Procedure Contributes to Tampa Bay Legacy of Success
The Tampa 2 procedure was created to be easy to replicate while achieving equal or better outcomes compared to the Cox-Maze IV, a widely used surgical intervention for AFib introduced in 2002. AFib is a cardiac condition noted by an irregular, frequently rapid heart rhythm and is typically treated with prescription, catheter-based and/or surgical interventions.
“We created the Tampa 2 procedure after years of collaboration between cardiology and cardiac surgery in conjunction with surgical pioneer Dr. James Cox (originator of the Cox-Maze IV procedure). This state-of-the-art technique has the potential to redefine surgical management of atrial fibrillation,” said Dr. Sherman.
Like its predecessor, the Tampa 2 creates scar tissue to block abnormal electrical signals that cause AFib but adds two lines of ablation (the method to create scar tissue) and has a different approach made possible by new tools and technology. The Tampa 2 achieved its namesake because of the two additional lines of ablation, developed by two Tampa physicians, all while tipping their hat to the success the Tampa Bay Buccaneers had with their Tampa 2 defense. More than 50 successful Tampa 2 procedures have been performed at St. Joseph’s Hospital, including the treatment of Brittany Williams, a patient of Dr. Sherman. Now the benefits of the Tampa 2 procedure extend beyond the walls of St. Joseph’s Hospital as Dr. Sherman and Dr. Makati host lectures across the country to train physicians on all aspects of AFib treatment. In addition, the two physicians presented their procedure and clinical findings at the 2024 International Society for Minimally Invasive Cardiothoracic Surgery annual meeting in Athens, Greece (see abstract here).”We are very proud that this novel procedure was developed here at BayCare and St. Joseph’s Hospital,” said Dr. Makati. “We are excited to continually offer new, lifesaving procedures to our community and beyond. This is only possible by the innovative spirit and collaborative efforts of our colleagues here in Tampa and around the world.” For more information about the Heart Institute at St. Joseph’s Hospital: https://baycare.org/locations/hospitals/st-josephs-hospital/services/heart-and-vascular About St. Joseph’s HospitalSt. Joseph’s Hospital, part of the BayCare Health System, is known for advanced medical technology and compassionate care. Its Centers of Excellence include the Heart and Vascular Institute, Cancer Institute, Stroke and Neuroscience Program, Robotic Surgery Program and Emergency/Trauma Department, which provides more emergency care than any other hospital in Tampa Bay. More than 70 specialties are represented among the medical team, including internal medicine, cardiovascular surgery and neurosurgery. The hospital was founded in 1934 by the Franciscan Sisters of Allegany. The 615-bed hospital is located at 3001 West Dr. Martin Luther King Jr. Boulevard in Tampa, Florida. For more information: BayCare.org/SJH.About BayCareBayCare is a leading not-for-profit health care system that connects individuals and families to a wide range of services at 16 hospitals and hundreds of other convenient locations throughout the Tampa Bay and central Florida regions. The system is West Central Florida’s largest provider of behavioral health and pediatric services and its provider group, BayCare Medical Group, is one of the largest in the region. BayCare’s diverse network of ambulatory services includes laboratories, imaging, surgical centers, BayCare Urgent Care locations, wellness centers and one of Florida’s largest home care agencies, BayCare HomeCare. BayCare’s mission is to improve the health of all it serves through community-owned, health care services that set the standard for high-quality, compassionate care. For more information visit BayCare.org.SOURCE BayCare Health System
Elucid Moves into New Headquarters in Preparation for Commercial Launch
BOSTON–(BUSINESS WIRE)–Elucid, a pioneering medical technology company providing physicians with imaging analysis software based on ground truth histology to support treatment of cardiovascular disease, today announced it has moved its headquarters to 399 Boylston Street in the heart of Boston’s Back Bay neighborhood. The move into the modern space comes […]
UltraSight Joins Butterfly Garden to Expand AI Real-Time Guidance Across Point of Care Ultrasound Devices
Leveraging Butterfly Network’s platform, UltraSight seeks to enable patients to access cardiac imaging with real-time AI guidance
TEL AVIV, Israel, June 27, 2024 /PRNewswire/ — Today, UltraSight, a digital health pioneer transforming cardiac imaging through the power of artificial intelligence, announced it has joined Butterfly Garden, an artificial intelligence (AI) Marketplace launched by Butterfly Network, Inc. (“Butterfly”) (NYSE: BFLY), a digital health company transforming care through the power of portable, semiconductor-based ultrasound technology and intuitive software.
UltraSight and Butterfly have partnered to increase patient access to cardiac care by enabling more healthcare professionals to perform cardiac ultrasound. Subject to regulatory approvals and authorizations, UltraSight aims to integrate and deploy its real-time AI guidance software on Butterfly’s imaging platform and build the software for use with Butterfly’s single-probe, whole-body handheld ultrasound system.
Cardiovascular disease (CVD) is the leading global cause of death, accounting for an estimated 18 million deaths yearly. Today patients face significant delays in receiving crucial cardiac testing due to a fragmented market full of system bottlenecks, in addition to a national shortage of expert sonographers.
UltraSight’s mission is to empower any medical professional, including novice users with no prior sonography experience, to confidently conduct echocardiographic examinations and capture diagnostic quality cardiac ultrasound images at the point of care. Achieving compatibility with the most prominent handheld ultrasound device companies in the market, such as Butterfly, facilitates the company’s goal of increasing access to cardiac care and reducing system bottlenecks for patients.
“Joining the Butterfly Garden marks an important moment in our mission to revolutionize cardiac care,” said Davidi Vortman, CEO of UltraSight. “By integrating our real-time AI guidance software with Butterfly’s cutting-edge ultrasound technology, we are poised to transform the landscape of cardiac imaging. This collaboration will empower healthcare professionals, regardless of their experience level, to perform accurate and timely cardiac ultrasound exams at the point of care. Together, we will break down existing barriers in cardiac care, ensuring that more patients receive the critical diagnostics they need, when and where they need it most.”
“We are thrilled to partner with UltraSight to bring their AI guidance software to Butterfly devices in an effort to mitigate the pressing issue of sonographer shortages, among other workforce challenges, impacting medical communities around the world,” said Darius Shahida, chief strategy officer of Butterfly Network. “UltraSight and Butterfly are jointly dedicated to making healthcare more efficient, effective and accessible through high-quality ultrasound that’s easy-to-use and globally available.”
In August 2023, Butterfly Network launched Butterfly Garden, allowing third-party developers access to its proprietary SDK and APIs to build new AI applications that work in conjunction with Butterfly’s imaging platform, bringing with it access to the largest point-of-care ultrasound customer base. UltraSight, with capabilities to provide more medical professionals with the ability to take high-quality diagnostic images of the heart, has the potential to close the gap between novice and skilled Butterfly ultrasound users, and is an ideal match for the program.
When paired with ultrasound devices, and following the appropriate regulatory clearance for each device, UltraSight’s underlying AI neural network can predict the position of the ultrasound probe relative to the heart based on the ultrasound video stream and guide the user on maneuvering the probe to capture diagnostic quality cardiac images.
UltraSight’s partnership with Butterfly follows a series of recent collaborations with other industry leaders such as Mayo Clinic and EchoNous. For more information about UltraSight, visit www.ultrasight.com. For more information about Butterfly, visit www.butterflynetwork.com.
About UltraSight
UltraSight’s mission is to make diagnostic imaging more accessible by empowering medical professionals to successfully acquire timely and accurate cardiac ultrasound images anywhere. UltraSight’s AI-driven software offers real-time guidance, making cardiac ultrasound accessible and efficient, which may lead to quicker diagnoses and improved patient care. In 2022, UltraSight won the Bristol Myers Squibb Improving Cardiovascular Disease Outcomes Challenge as the most “innovative cardiac technology.” Additionally, the company was awarded a patent for its real-time guidance solution for ultrasound devices. UltraSight’s software has FDA 510(k) Clearance, is UKCA and CE Marked, and has Israeli AMAR Clearance to assist medical professionals in performing cardiac ultrasound scans. For more news and information, visit our website or follow UltraSight on LinkedIn and Twitter
About Butterfly Network
Founded by Dr. Jonathan Rothberg in 2011, Butterfly Network is a digital health company with a mission to democratize medical imaging by making high-quality ultrasound affordable, easy-to-use, globally accessible, and intelligently connected, including for the 4.7 billion people around the world lacking access to ultrasound. Butterfly created the world’s first handheld single-probe, whole-body ultrasound system using semiconductor technology, Butterfly iQ. The company has continued to innovate, leveraging the benefits of Moore’s Law, to launch its second-generation Butterfly iQ+ in 2020, and third-generation iQ3 in 2024 – each with increased processing power and performance enhancements. The disruptive technology has been recognized by TIME’s Best Inventions, Fast Company’s World Changing Ideas, CNBC Disruptor 50, and MedTech Breakthrough Awards, among other accolades. With its proprietary Ultrasound-on-Chip™ technology, intelligent software, and educational offerings, Butterfly is paving the way to mass adoption of ultrasound for earlier detection and remote management of health conditions around the world. Butterfly devices are commercially available to trained healthcare practitioners in areas including, but not limited to, parts of Africa, Asia, Australia, Europe, the Middle East, North America and South America; to learn more about available countries, visit: www.butterflynetwork.com/choose-your-country.
SOURCE UltraSight
VenoStent Completes $20 Million Series A with $4 Million Investment from Norwest Venture Partners
Company Builds on Momentum with New $3.6 Million NIH SBIR Grant to Fund U.S. Clinical Trial of Breakthrough Medical Device Showing Promise in Improving Dialysis Patient Outcomes
HOUSTON, June 27, 2024 /PRNewswire/ — VenoStent, Inc., a clinical-stage medical device company developing a novel therapeutic device for improving dialysis patient outcomes, announced today that it closed an additional $4 million from Norwest Venture Partners to round out its Series A financing at $20 million with Good Growth Capital and IAG Capital Partners co-leading the deal. In addition, the company was awarded a $3.6 million Small Business Innovation Research (SBIR) Phase II Grant by the National Institutes of Health (NIH), which will help fund its multi-center, 200-patient, randomized controlled trial (RCT) in the US.
Norwest General Partner Dr. Zack Scott and Investor Dr. Ehi Akhirome are joining the company as board observers. Dr. Scott and Dr. Akhirome bring deep medtech expertise from previous experience as a surgeon and physician scientist, respectively.
“2024 has been a momentous year for VenoStent so far. In the span of a few months, we initiated our first clinical sites, enrolled the first patients in our large RCT and closed our Series A with Norwest,” said Tim Boire, Ph.D., VenoStent CEO and co-founder. “We also received the NIH grant, which enables us to execute our trial with the highest degree of quality and rigor to make it as scientifically robust and impactful to patients as possible. Each of these are major company milestones that collectively represent many years of intensive and fruitful R&D and collaboration. These recent milestones will propel our company forward to an exciting next phase.”
As a clinical-stage therapeutic medical device company, VenoStent has developed a bioabsorbable perivascular wrap, SelfWrap, that goes around arteriovenous (AV) access sites at the time of AV fistula creation surgery. The bioabsorable wrap is intended to accelerate the usability and increase the durability of the fistula sites for chronic kidney disease (CKD) patients requiring hemodialysis. SelfWrap uses the body’s own healing mechanisms to mimic the arterial environment in veins, which experience a 10x increase in pressure and flow during AV creation and causes the veins to become unusable in dialysis.
In May 2023, SelfWrap was approved by the U.S. Food and Drug Administration to begin its U.S. Investigational Device Exemption (IDE) study, SAVE-FistulaS: The SelfWrap-Assisted ArterioVEnous Fistulas Study. The study is designed to show how SelfWrap can improve clinical outcomes for CKD patients requiring hemodialysis. Based on the compelling results from the company’s first-in-human clinical trial, the FDA granted SelfWrap a Breakthrough Device Designation in May 2022.
“Over half a million people in the U.S. rely on hemodialysis to survive and require an arteriovenous fistula creation surgery in order to receive the treatment. However, the AV fistula procedure has a one-year failure rate of more than 60%, which significantly impacts patients’ survival rates and quality of life,” said Norwest’s Dr. Scott. “VenoStent’s groundbreaking technology for AV fistula formation, SelfWrap, has the potential to significantly improve these odds. We look forward to working with the VenoStent team as it proves the efficacy of this breakthrough technology in order to improve the lives of hundreds of thousands of CKD patients.”
“Norwest’s investment is tremendous validation for VenoStent, and we are thrilled to have both Zack and Ehi joining the company’s board,” said VenoStent COO and Co-Founder, Geoffrey Lucks. “Zack and Ehi have extensive knowledge in our space, and their added value will match the capital and cache of Norwest dollar-for-dollar.”
About VenoStentVenoStent, Inc. (www.venostent.com) is a therapeutic medical device company developing a bioabsorbable wrap, SelfWrap, to transform outcomes in vascular surgery, starting with hemodialysis access. The device is intended to improve the usability and durability of arteriovenous fistulas (AVFs), the artery-vein connections that are surgically created in the arms of CKD patients to enable life-saving dialysis treatments. SelfWrap has been finely tuned through a decade of development to provide optimal mechanical support, improving hemodynamics and outward vein growth, and ultimately leading to improved quality and length of life for end-stage renal disease (ESRD) patients. Headquartered in Houston, Texas, VenoStent is venture backed by Good Growth Capital, IAG Capital Partners and Norwest Venture Partners, amongst others.
About Norwest Venture PartnersNorwest Venture Partners is a global venture and growth equity investment firm managing more than $15.5 billion in capital. Since its inception, Norwest has invested in more than 700 companies and currently partners with more than 230 companies in its venture and growth equity portfolio. The firm invests in early- to late-stage businesses across key sectors with a focus on enterprise, consumer and healthcare. The Norwest team offers a deep network of connections, extensive operating experience, and a wide range of impactful services to help CEOs and founders scale their businesses. Norwest has offices in Menlo Park and San Francisco, Calif.; Mumbai, India; and Tel Aviv, Israel. For more information, please visit www.nvp.com.
SOURCE VenoStent
Bioxodes meets first Phase 2a patient enrollment milestone with BIOX-101 in intracerebral hemorrhagic stroke
First-in-class drug candidate evaluated in first eight patients Gosselies (Belgium), June 27, 2024 – Bioxodes SA, a clinical stage biopharmaceutical company developing novel therapies for the prevention and treatment of thrombotic and inflammatory diseases, announces today that it has enrolled the first eight of 32 patients in a Phase 2a clinical study of its lead asset BIOX-101 (Ir-CPI). The study is evaluating BIOX-101 in intracerebral hemorrhagic stroke (ICH), a devastating condition for which there is currently no available treatment. Reaching this first milestone allows Bioxodes to conduct an initial analysis of pharmacokinetic and pharmacodynamic data to evaluate the dose-response and preliminary clinical proof-of concept of the therapeutic candidate, as well as safety in this patient population. “For the first time, we are able to assess the preliminary results of BIOX-101 in ICH patients, who up until now have had very few treatment options. Analysis of these first 8 patients will yield preliminary proof-of-concept data for BIOX-101, a first-in-class drug candidate derived from a protein found in the saliva of the tick 1, designed to prevent the harmful secondary brain injuries that occur after a hemorrhagic stroke,” said Marc Dechamps, Chief Executive Officer at Bioxodes. The study, conducted in 10 stroke units in Belgium and led by Prof Robin Lemmens, a world-leading stroke authority and head of the clinic at the University Hospital Leuven, aims to enroll 32 patients aged 18 and above, with 24 receiving BIOX-101, and 8 standard-of-care treatment. The trial is a randomized, open-label proof-of-concept study, and will evaluate the safety and tolerability of BIOX-101 in patients with spontaneous ICH, while also generating preliminary data on secondary efficacy objectives. All patients will be monitored for at least one year to evaluate the impact of the treatment on long-term functional outcomes. Interim results for the first 16 patients are expected by the fourth quarter of 2024. BIOX-101 prevents blood clot formation without increasing the risk of further bleeding. Moreover, by inhibiting the activation of neutrophils, a type of white blood cell that often act as the first responders of the inflammatory system, it also prevents the acute neuroinflammatory events associated with ICH. BIOX-101 is also in early development as a platform for a series of other indications, including ischemic stroke and other thrombo-inflammatory diseases. KEY FACTS ABOUT STROKE:* 15 million people worldwide suffer from a stroke each year, * Of those, 5 million die, another 5 million are left severely disabled* Stroke is the leading cause of disability among neurological conditions worldwide, according to the Global Burden of Disease Study 2021 (Lancet Neurology May 2024).* Two types: ischemic stroke (caused by a blood clot) and hemorrhagic stroke (ruptured vessel)* Hemorrhagic stroke makes up 13% of all cases, but causes 40% of deaths* Intracerebral hemorrhagic stroke (ICH) is by far the most common type of hemorrhagic stroke * For hemorrhagic stroke, there is little other to do than surgery, with poor outcomes* Bioxodes has applied for an orphan disease designation for BIOX-101 in the US and EU. Being awarded the designation could accelerate the regulatory approval process. 1 Ixodes ricinus About Bioxodes Bioxodes is a clinical stage biopharmaceutical company developing novel therapies for the prevention and treatment of thrombotic and inflammatory diseases. Since its founding in 2013, Bioxodes has developed its lead asset BIOX-101, a first-in-class drug candidate aimed at patients with thrombo-inflammatory disease. BIOX-101’s unique mechanism of action is the foundation of an innovative pipeline of drug candidates for the prevention of (thrombo)inflammatory diseases. The company, which is based in the biopark of Gosselies near Brussels in Belgium, has so far secured €34 million in funding from Belgian investment funds and business angels, including €12 million in non-dilutive funding from the Wallonia region. Worldwide, Bioxodes holds both granted and pending patents associated with BIOX-101. http://www.bioxodes.com/ For more information please contact: HEAD OFFICESBioPark Charleroi-Bruxelles SudRue Santos-Dumont, 16041 Gosselies, Belgium+32 496 590354investment@bioxodes.com MEDIA RELATIONSAlexandra Schiettekatte alexandra.alicato@outlook.com+32 476 65 04 38COHESION BUREAUEU MEDIA RELATIONSSophie Baumontsophie.baumont@cohesionbureau.comINVESTOR RELATIONSGiovanni Ca’ Zorzigiovanni.cazorzi@cohesionbureau.com
Attachment
20240627 Bioxodes PR first 8 patients (final) EN
Tempus Receives U.S. FDA 510(k) Clearance for Tempus ECG-AF, an AI-based Algorithm that Identifies Patients at Increased Risk of AFib
CHICAGO–(BUSINESS WIRE)–Tempus AI, Inc. (NASDAQ: TEM), a leader in artificial intelligence and precision medicine, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Tempus ECG-AF device that uses AI to help identify patients who may be at increased risk of atrial fibrillation/flutter […]
American College of Radiology Launches First Medical Practice Artificial Intelligence Quality Assurance Program
ARCH-AI Can Help Radiology Sites Safely and Effectively Implement AI in Daily Practice
RESTON, Va., June 26, 2024 /PRNewswire/ — The American College of Radiology® (ACR®) today launched the ACR Recognized Center for Healthcare-AI (ARCH-AI), the first national artificial intelligence quality assurance program for radiology facilities.
The program, built on best practices, outlines expert consensus-based building blocks of infrastructure, processes and governance in AI implementation in real-world practice.
By working toward, and attesting to, compliance within the tenets of the program, participation in ARCH-AI can help radiology practices provide safe and effective implementation of AI products and help radiologists provide better patient care.
“AI is different from previous technologies,” said Christoph Wald, MD, PhD, MBA, FACR, vice chair of the ACR Board of Chancellors and chair of the ACR Commission on Informatics. “Even a U.S. Food and Drug Administration-cleared AI product must be tested locally to ensure it works safely and as intended. Practice leaders must put safeguards in place to maximize the benefit of AI products while minimizing risk; ARCH-AI is a low-cost, efficient system to help sites do that.”
ARCH-AI site recognition criteria include:
Establishing an interdisciplinary AI governance group.
Maintaining an inventory of AI algorithms with detailed documentation.
Ensuring adherence to security and compliance measures.
Engaging in diligent review and selection of AI algorithms.
Documenting use cases and training procedures.
Monitoring algorithm performance, including safety and effectiveness.
Contributing to the “Assess-AI” central AI registry for performance benchmarking.
“ARCH-AI can help radiology practices structure QA processes that help them plan for what can go wrong, including the development of good AI governance practices, acceptance testing and effectiveness monitoring of AI products to ensure they continue to function as expected over time,” said Keith J. Dreyer, DO, PhD, FACR, ACR Data Science Institute® (DSI®) chief science officer.
Radiology practices that complete the ARCH-AI process will receive an ACR Recognition badge to display in their waiting rooms and lobbies to demonstrate to their communities, patients, payers and referring physicians that they are committed to integrating AI in a safe, responsible manner that allows them to provide the best possible modern healthcare.
“ARCH-AI provides a great blueprint and assists radiology facilities with AI implementation,” said Bibb Allen Jr., MD, FACR, ACR DSI chief medical officer. “DSI also offers AICentral.org, a database of FDA-cleared imaging AI products, and Assess-AI, the ACR’s AI registry that can support local AI acceptance testing and monitoring. These ACR programs can help practices meet ARCH-AI standards and receive meaningful guidance and assistance in implementing AI in a safe and effective manner for their patients.”
SOURCE American College of Radiology
Rep. Hillary Scholten and the National Blood Clot Alliance Award Corewell Health Butterworth Hospital as a VTE Center of Excellence
GRAND RAPIDS, Mich., June 26, 2024 /PRNewswire/ — On June 24, 2024, Rep. Hillary Scholten (MI-03) joined members of the National Blood Clot Alliance “NBCA” (www.stoptheclot.org) to present NBCA’s inaugural Venous Thromboembolism (VTE) Center of Excellence (COE) award to Corewell Health Grand Rapids Hospitals – Butterworth Hospital. This award recognizes the hospital’s outstanding commitment to providing the highest standard of care, patient education, and follow-up treatment for VTE patients.
Continue Reading
Rep. Hillary Scholten (MI-03) (center) presents the inaugural NBCA VTE Center of Excellence Award to the Corewell Health team. Pictured from left to right: Glen VanOtteren, Erin VanDyke, PA-C, MPAS, Michael Knox, MD, and Trevor Cummings, MD, FACEP.
Rep. Scholten stated, “Our friends, families, neighbors, and the wider community can trust the commitment to excellence demonstrated by Corewell’s clinicians. These incredible providers ensure that all patients, not just some, will receive the highest standard of care when being diagnosed and treated for VTE. I was delighted to submit a FY25 appropriations request to fund a nationwide public awareness campaign and to train healthcare providers to recognize the symptoms of blood clots. West Michigan is leading the way in addressing this public health crisis, setting a model for both the Great Lakes State and our nation.”
Dr. Michael Knox, MD, FACR, and Co-Director of the Corewell PERT Program, accepted the award on behalf of Corewell. He stated, “I am honored to accept this award on behalf of my team and thank NBCA for this distinction. We appreciate Rep. Scholten’s acknowledgement of the important work we are doing. Tackling this public health crisis will take all of us working together — hospitals, clinicians, patients, and Members of Congress.”
Justin Crockett, NBCA Board Member and Chair of the NBCA VTE COE Committee, added, “Development of this VTE COE designation has been a long journey. We are very proud to present this award and to start reducing the incidence and mortality of VTE, which claims the lives of 100,000 people each year from all age groups and walks of life. We are especially grateful for Congresswoman Scholten’s support and in recognizing the toll VTE takes on both the citizens of Michigan and the entire nation. We also thank her for supporting NBCA’s $5 million federal appropriations request directing funding to the Centers for Disease Control and Prevention’s (CDC) Division of Blood Disorders and Genomics for a national VTE education, awareness, and prevention program for both the public and clinicians.”VTE (venous blood clots) claims the lives of 100,000 Americans each year while impacting another 900,000 individuals. It is the third leading cause of cardiovascular death and the second cause of sudden unexpected death. It kills more people each year than HIV/AIDS, breast cancer and motor vehicle accidents combined. Nearly half of all VTE cases occur in a hospital or post-hospital setting, yet, according to the CDC, 70% of these cases are preventable. It is estimated that the annual cost to the US healthcare system is $10 billion per annum.About the National Blood Clot Alliance The National Blood Clot Alliance, established in 2003, is the nation’s leading nonprofit, patient advocacy organization dedicated to advancing the prevention, early diagnosis, and successful treatment of life-threatening blood clots, such as deep vein thrombosis (DVT) and pulmonary embolism (PE), DVT +PE=VTE. Each year NBCA provides blood clot information, resources, and support to more than 3 million people. For more information about NBCA please contact [email protected] or visit www.stoptheclot.org.About Corewell Health™People are at the heart of everything we do, and the inspiration for our legacy of outstanding outcomes, innovation, strong community partnerships, philanthropy and transparency. Corewell Health is a not-for-profit health system that provides health care and coverage with an exceptional team of 65,000+ dedicated people—including more than 12,000 physicians and advanced practice providers and more than 15,500 nurses providing care and services in 21 hospitals, 300+ outpatient locations and several post-acute facilities—and Priority Health, a provider-sponsored health plan serving more than 1.3 million members. Through experience and collaboration, we are reimagining a better, more equitable model of health and wellness. For more information, visit www.corewellhealth.org.About Rep. Hillary Scholten:Congresswoman Hillary J. Scholten is honored to serve the people of Michigan’s Third Congressional District, a diverse district that is anchored by the urban centers of Grand Rapids, Muskegon, and Grand Haven, and includes suburbs, farmland, and miles of beautiful Lake Michigan shoreline.Congresswoman Scholten is a fourth-generation West Michigander. For more information visit www.hillaryscholten.com.SOURCE National Blood Clot Alliance
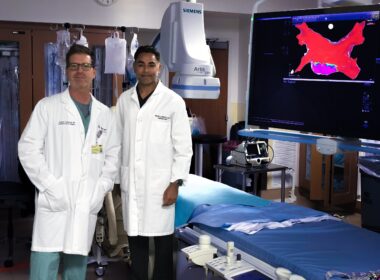
Fort Sanders Regional Medical Center First in Tennessee to Adopt New Cardiac Angiogram Technology
KNOXVILLE, Tenn., June 26, 2024 /PRNewswire/ — Fort Sanders Regional Medical Center, a member of Knoxville-based Covenant Health, was the first hospital in the state of Tennessee to adopt revolutionary diagnostic technology that provides clinicians with a patient’s physiology results in just a few minutes.
Coronary heart disease is one of the top killers of adults in the U.S. and particularly the Southeast. Quick diagnosis and intervention are key to treating cardiac patients who are suffering from heart disease, heart attack, or other events causing blockages in arteries that can be life-threatening.
Fort Sanders Regional’s cardiac catheterization lab is staffed around the clock, 24/7, treating both heart patients who have been admitted to the hospital and those who are having outpatient procedures.
When a patient presents with a potential artery blockage, the heart team at Fort Sanders Regional can use a new tool that shows doctors exactly what percentage of an artery is blocked. The CathWorks FFRangio ® System is a computer-based technology that uses routine angiograms (images) of a patient’s heart structure to create a 3D model showing the location and degree of any blockages and their coronary arteries. This helps providers more quickly diagnose and intervene as needed, including determining whether the patient needs a stent and where the stent should be placed in the heart structure.
Fractional flow reserve (FFR) is a diagnostic measurement that evaluates the physiologic impact of coronary artery narrowing due to cholesterol plaque. It is an important part of the decision-making process to decide if a stent placement is indicated and the length of the stent when managing patients with coronary artery disease (CAD).
In contrast to traditional FFR, the FFRangio System combines artificial intelligence (AI) and advanced computational science to obtain quick and reliable FFRangio values from routine angiograms, eliminating the need for drug stimulation and invasive pressure wires. The system provides physicians intraprocedural FFRangio values for all coronary arteries.
Josh Todd, MD, interventional cardiologist at Fort Sanders Regional, said, “With this new tool, interventional cardiologists can perform clinical assessments quicker, more comprehensively, without medications, and in a way that’s seamless and more efficient to the patient. This cutting-edge technology will transform how cardiovascular disease is diagnosed and treated.”
To view the full release, visit https://www.covenanthealth.com/blog/cathworks-technology/.
SOURCE Covenant Health