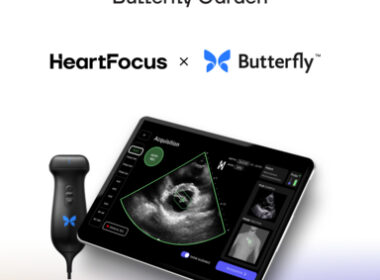
June 24, 2024 07:00 AM Eastern Daylight Time BORDEAUX, France–(BUSINESS WIRE)–HeartFocus, the revolutionary, AI-driven heart exam software by DESKi, a provider of automated, accurate, and data-driven image analysis tools today announced it has joined Butterfly Garden, an artificial intelligence (AI) Marketplace launched by Butterfly Network, Inc., (“Butterfly”) (NYSE: BFLY) a digital health […]
Other News
Heart Hospital of New Mexico at Lovelace Medical Center and GE HealthCare Collaborate to Install the First Allia IGS Pulse Electrophysiology Lab in the United States for Minimally Invasive Cardiovascular Procedures
ALBUQUERQUE, N.M.–(BUSINESS WIRE)–Today Heart Hospital of New Mexico at Lovelace Medical Center (HHNM) and GE HealthCare (Nasdaq: GEHC) announced HHNM as the first location in the United States to install GE HealthCare’s latest Allia Image Guided System (IGS) Pulse, which was designed to provide exceptional image quality and improve workflow for the diagnosis and […]
MemorialCare Heart & Vascular Institute at Long Beach Medical Center is First Hospital in Southern California to Perform Transcatheter Aortic Valve Implantation with Navitor Titon 35mm Valve
This is the first 35mm size valve implantation giving those with large native aortic valves at high-risk a minimally invasive option – avoiding open heart surgeryLONG BEACH, Calif., June 21, 2024 /PRNewswire/ — Yesterday, MemorialCare Heart & Vascular Institute at Long Beach Medical Center was the first hospital in Southern California to implant a 35mm Abbott Titan Navitor Valve during a Transcatheter Aortic Valve Implantation (TAVI) procedure. The TAVI procedure is crucial for patients who have severe aortic stenosis and are at extreme risk for open-heart surgery. This offers a minimally invasive approach which shortens hospital stays, reduces risk of infection and lessens recovery time.
Continue Reading
Titan Navitor Valve Team from MemorialCare Heart and Vascular Institute at Long Beach Medical Center
“We are very proud of Dr. Anthony Chyou and Dr. Ali Khoynezhad for leading us through this transformational time of our heart and vascular institute,” says David Shavelle, M.D., FACC, chief of cardiology, MemorialCare Heart & Vascular Institute; medical director, adult cardiology & interventional lab, MemorialCare Heart & Vascular Institute at Long Beach Medical Center. “We have always taken pride in creating an environment where our patients are given personalized treatment. The Navitor Titan 35mm Valve allowed us to provide a minimally invasive treatment option to a high-risk patient with severe aortic stenosis.”
The Navitor valve is one of the newest heart valve devices approved by the U.S. Food and Drug Administration for TAVI, also known as Transcatheter Aortic Valve Replacement (TAVR). This device is different from other heart valve replacement valves on the market due to improved blood flow (hemodynamic) performance and ease of use.
“The intuitive design of the Navitor TAVI System allows more patients who normally would not be viable candidates for a Transcatheter Aortic Valve Replacement procedure access to a valve replacement,” says Optum partnering physician Anthony Chyou, M.D., who is also an interventional cardiologist at MemorialCare Heart & Vascular Institute at Long Beach Medical Center. “Patients with large native aortic valves and severe aortic stenosis valve disease may not have been able to receive treatment as prior TAVI valves were not large enough to accommodate their anatomy.”Heart valve disease limits the ability of the heart to pump blood through the entire body. The Navitor TAVI System has been tested and shown to greatly improve the blood flow of patients with symptomatic severe aortic stenosis, helping their hearts resume to normal, healthy blood flow performance quickly.”MemorialCare is dedicated to state-of-the-art, patient-centric care to our community, and this ‘first in the region’ procedure is a testament to our continued commitment to excellent patient care,” says Ali Khoynezhad, M.D., director, aortic and arrythmia surgery, MemorialCare Heart & Vascular Institute at Long Beach Medical Center.The Navitor TAVI System has been rigorously tested and proven to yield positive 30-day outcomes in patients with symptomatic severe aortic stenosis, including a 0% chance of moderate or severe paravalvular leak (PVL), 1.9% chance of all-cause mortality, 1.9% chance of disabling stroke, and 4.2% chance of major vascular complications, proving its remarkable safety and effectiveness in TAVR procedures.About MemorialCare Long Beach Medical Center:MemorialCare Long Beach Medical Center is a member of MemorialCare, a not-for-profit, integrated healthcare system. Long Beach Medical Center has been providing the community with compassionate, quality health care for more than 100 years. At the forefront of specialized care, research, and education, Long Beach Medical Center uses the most advanced healthcare technologies, including pioneering surgical systems like – ExactechGPS® and ExcelsiusGPS®. Recognized among the top 3% of all California acute care hospitals, Long Beach Medical Center is recognized as “Best Hospital” for OBGYN and Orthopedics and ranked regionally in the Los Angeles Metro Area by U.S. News & World Report and earned Magnet® recognition for nursing excellence. With premier centers dedicated to cancer, heart, rehabilitation, orthopedics, neurosciences, and trauma, physicians and surrounding hospitals continually refer to its accredited programs. For more information, visit memorialcare.org/LongBeach.SOURCE MemorialCare Long Beach Medical Center
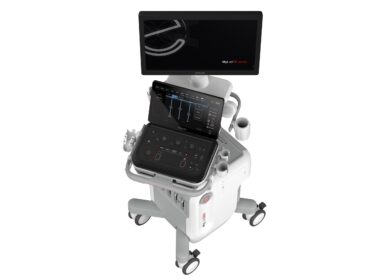
At the SIRM Congress 2024 in Milan, ESAOTE presents the brand-new MyLab™E80, an E-series ultrasound device designed for professionals dealing with more complex clinical cases
The unveiling took place at the Esaote stand (I-03), where industry professionals could find out more about the features and performance of the new system, throughout the Congress. MILAN, June 21, 2024 /PRNewswire/ — Esaote – a leading Italian company in the field of medical imaging – has unveiled its new MyLab™E80 ultrasound device to the world of radiology. The unveiling took place at the 51st SIRM Congress 2024 (Allianz MiCo Convention Center, Milan – Stand I-03), the first time the three scientific societies in the field of radiology (SIRM, AIMN, AIRO) have come together for their first joint Congress, entitled “The Next Generation”.
Continue Reading
The new Esaote MyLab™E80 ultrasound device
Another step forward in terms of innovation, MyLab™E80 is the first ultrasound system in Esaote’s brand-new E series. The ‘E’ stands for Expertise, referring to a series of devices designed for professionals who deal with more complex clinical cases, where diagnostic certainty and clinical efficiency play a decisive role.
This ultrasound scanner guarantees intuitive workflow management, with advanced A.I.-powered automation features to speed up repetitive operations and simplify complex functions. Doctors can therefore conduct more thorough examinations based on images with a very high level of detail. The system also comes with fusion imaging technology, for the assessment of liver disease and breast lesions.
MyLab™E80 integrates the Augmented Insight™ functions of the MyLab™X90 and provides users with options of a conventional or touchscreen control panel, in line with the innovative selection launched in the A Series. “The agile and fast MyLab™E80 and its versatile and mobile advanced features embodies our new vision as part of our ongoing commitment to providing high-quality healthcare. We have therefore improved the reproducibility, efficiency and diagnostic accuracy of our ultrasound scanners,” stated Mariagrazia Bella, Marketing Director for Italy. “MyLab™E80 is designed to adapt to any healthcare environment. With five probe connectors and custom configurations, it’s ideal for both routine and advanced examinations. The battery offers autonomous scanning at full power, while the modern touch panel simplifies cleaning procedures, ensuring adaptability even in the most demanding environments.”MyLab™E80 completes the renewal of the range of ultrasound scanners developed by Esaote, alongside the MyLab™X90, MyLab™A70 and MyLab™A50. In line with its new brand identity and slogan of “Health with Care”, Esaote confirms its mission and the sense of care and empathy with which it approaches its everyday work, to improve people’s well-being. www.esaote.com Esaote Group Leader in medical imaging (ultrasound, MRI, software to manage the diagnostic process). At the end of 2023, the Group had 1,250 employees, half of whom in Italy. With facilities in Genoa and Florence, and its own production and research units in Italy and the Netherlands, Esaote maintains a presence in over 100 countries. www.esaote.com© Copyright Esaote 2024Photo – https://mma.prnewswire.com/media/2442819/Esaote_SpA.jpgLogo – https://mma.prnewswire.com/media/2323931/4771337/ESAOTE_Logo.jpg
Acasti Announces Year-End 2024 Financial Results, Provides Business Update
Patient Enrollment in Pivotal STRIVE-ON Phase 3 Safety Trial for GTX-104 On-Track for Potential NDA Submission in 1H Calendar 2025Projected Cash Runway into Second Calendar Quarter 2026 PRINCETON, N.J., June 21, 2024 (GLOBE NEWSWIRE) — Acasti Pharma Inc. (Nasdaq: ACST) (Acasti or the Company), a late-stage, biopharma company advancing GTX-104, its novel injectable formulation of nimodipine that addresses high unmet medical needs for a rare disease, aneurysmal subarachnoid hemorrhage (aSAH), today announced financial results and business highlights for the year ended March 31, 2024. “During the past year we continued to execute our focused strategy around our biggest value driver program GTX-104 and its pivotal Phase 3 STRIVE-ON safety trial (the STRIVE-ON trial–NCT05995405),” said Prashant Kohli, CEO of Acasti. “Since initiating STRIVE-ON and dosing the first patient last October, enrollment has proceeded steadily, and we believe the trial is on track for a potential NDA submission to the FDA in the first half of calendar 2025. With our balance sheet enhanced by the $7.5 million private placement secured in September 2023, and disciplined execution of the strategic realignment plan we announced in May 2023, our cash runway is expected to extend into the second calendar quarter of 2026, well beyond our planned submission of the GTX-104 NDA.” 2024 Corporate Highlights Overview of STRIVE-ON trial, a prospective, open-label, randomized (1:1 ratio), parallel group trial of GTX-104 compared with oral nimodipine, in patients hospitalized for aSAH, presented as a poster at the 2024 International Stroke ConferenceEnrollment of STRIVE-ON trial on track for potential NDA submission to FDA anticipated in the first half of calendar 2025Conducted a meeting of STRIVE-ON trial investigators in April 2024; the in-person/virtual meeting provided an excellent opportunity to energize the principal investigators from all sites, engage in an interactive discussion about the trial inclusion/exclusion criteria, and to review key procedures to ensure quality data collectionIn October 2023 hosted a Key Opinion Leader Event GTX-104: A Potential New Treatment Standard for Rare and Life-Threatening aneurysmal Subarachnoid Hemorrhage (aSAH)Completed $7.5 million private placement equity financing led by ADAR1 Partners, LP in September 2023Presented a pharmacokinetic comparison of GTX-104 with oral nimodipine at the 2023 Neurocritical Care Society annual meeting in August 2023. Fiscal Year 2024 Financial Results The Company reported a net loss of $12.9 million, or $1.35 loss per share, for the year ended March 31, 2024, a decrease of $29.5 million from the net loss of $42.4 million or $5.71 per share for the year ended March 31, 2023. The decrease in net loss was primarily due to asset impairments, net of income tax benefit, totaling $25.3 million during the year ended March 31, 2023 that did not recur during the year ended March 31, 2024, the impact of the Company’s strategic realignment in May 2023 to align the organizational and management cost structure to prioritize resources to GTX-104, and the change in fair value of the Company’s derivative warrant liabilities that was primary attributable to an increase in stock price. Research and development expenses for the year ended March 31, 2024 were $4.7 million, compared to $10.0 million for the year March 31, 2023. The decrease from the prior year period was mainly attributable to the Company’s strategic realignment in May 2023, which resulted in reduced clinical development and salaries and benefits expenses. General and administrative expenses were $6.4 million for the year ended March 31, 2024, a decrease of $1.2 million from $7.6 million for the year ended March 31, 2023. The decrease was primarily a result of decreased salaries and benefits due to a reduction in general and administrative headcount as a result of the restructuring and reorganization of our management structure offset by increased legal, tax, accounting and other professional fees related to the restructuring and private placement. At March 31, 2024 the Company had cash and cash equivalents of $23.0 million, as compared to $27.9 million as of March 31, 2023. The Company believes it has sufficient cash to support operations into the second calendar quarter of 2026. About aneurysmal Subarachnoid Hemorrhage (aSAH) aSAH is bleeding over the surface of the brain in the subarachnoid space between the brain and the skull, which contains blood vessels that supply the brain. A primary cause of such bleeding is the rupture of an aneurysm. Approximately 70% of aSAH patients experience death or dependence, and more than 30% die within one month of hemorrhage. Approximately 50,000 patients in the United States are affected by aSAH per year, based on market research. Outside of the United States, annual cases of aSAH are estimated at approximately 60,000 in the European Union, and approximately 150,000 in China. About the Acasti Asset Portfolio GTX-104 is a clinical stage, novel, injectable formulation of nimodipine being developed for intravenous (IV) infusion in aSAH patients to address significant unmet medical needs. The unique nanoparticle technology of GTX-104 facilitates aqueous formulation of insoluble nimodipine for a standard peripheral IV infusion. GTX-104 provides a convenient IV delivery of nimodipine in the Intensive Care Unit potentially eliminating the need for nasogastric tube administration in unconscious or dysphagic patients. Intravenous delivery of GTX-104 also has the potential to lower food effects, drug-to-drug interactions, and eliminate potential dosing errors. Further, GTX-104 has the potential to better manage hypotension in aSAH patients. GTX-104 has been administered in over 150 healthy volunteers and was well tolerated with significantly lower inter- and intra-subject pharmacokinetic variability compared to oral nimodipine. The addressable market in the United States for GTX-104 is estimated to be about $300 million, based on market research. GTX-102 is a novel, concentrated oral-mucosal spray of betamethasone intended to improve neurological symptoms of Ataxia-Telangiectasia (A-T), for which there are currently no FDA-approved therapies. GTX-102 is a stable, concentrated oral spray formulation comprised of the gluco-corticosteroid betamethasone that, together with other excipients can be sprayed conveniently over the tongue of the A-T patient and is rapidly absorbed. The further development of GTX-102 has been deprioritized in favor of our focus on development of GTX-104. It is also possible that we may out-license or sell our GTX-102 drug candidate. GTX-101 is a non-narcotic, topical bio-adhesive film-forming bupivacaine spray designed to ease the symptoms of patients suffering with postherpetic neuralgia (“PHN”). GTX-101 is administered via a metered-dose of bupivacaine spray and forms a thin bio-adhesive topical film on the surface of the patient’s skin, which enables a touch-free, non-greasy application. It also comes in convenient, portable 30 ml plastic bottles. Unlike oral gabapentin and lidocaine patches, we believe that the biphasic delivery mechanism of GTX-101 has the potential for rapid onset of action and continuous pain relief for up to eight hours. No skin sensitivity was reported in a Phase 1 trial. The further development of GTX-101 has been deprioritized in favor of our focus on development of GTX-104. It is also possible that we may out-license or sell our GTX-101 drug candidate. About Acasti Acasti is a late-stage biopharma company with drug candidates addressing rare and orphan diseases. Acasti’s novel drug delivery technologies have the potential to improve the performance of currently marketed drugs by achieving faster onset of action, enhanced efficacy, reduced side effects, and more convenient drug delivery. Acasti’s lead clinical assets have each been granted Orphan Drug Designation by the FDA, which provides seven years of marketing exclusivity post-launch in the United States, and additional intellectual property protection with over 40 granted and pending patents. Acasti’s lead clinical asset, GTX-104, is an intravenous infusion targeting aneurysmal Subarachnoid Hemorrhage (aSAH), a rare and life-threatening medical emergency in which bleeding occurs over the surface of the brain in the subarachnoid space between the brain and skull. For more information, please visit: www.acasti.com. Forward-Looking Statements Statements in this press release that are not statements of historical or current fact constitute “forward-looking statements” within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, as amended, Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, and “forward-looking information” within the meaning of Canadian securities laws (collectively, “forward-looking statements”). Such forward looking statements involve known and unknown risks, uncertainties, and other factors that could cause the actual results of Acasti to be materially different from historical results or from any future results expressed or implied by such forward-looking statements. In addition to statements which explicitly describe such risks and uncertainties, readers are urged to consider statements containing the terms “believes,” “belief,” “expects,” “intends,” “anticipates,” “estimates”, “potential,” “should,” “may,” “will,” “plans,” “continue”, “targeted” or other similar expressions to be uncertain and forward-looking. Readers are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date of this press release. The forward-looking statements in this press release, including statements regarding the Company’s anticipated cash runway, the timing of the planned NDA submission with the FDA in connection with the Company’s STRIVE-ON trial, GTX-104’s commercial prospects, GTX-104’s potential to bring enhanced treatment options to patients suffering from aSAH, and the anticipated benefits and future development, license or sale of the Company’s other drug candidates are based upon Acasti’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, including, without limitation: (i) the success and timing of regulatory submissions of the Phase 3 safety trial for GTX-104; (ii) regulatory requirements or developments and the outcome and timing of the proposed NDA application for GTX-104; (iii) changes to clinical trial designs and regulatory pathways; (iv) legislative, regulatory, political and economic developments; and (v) actual costs associated with Acasti’s clinical trials as compared to management’s current expectations. The foregoing list of important factors that could cause actual events to differ from expectations should not be construed as exhaustive and should be read in conjunction with statements that are included herein and elsewhere, including the risk factors detailed in documents that have been and are filed by Acasti from time to time with the Securities and Exchange Commission and Canadian securities regulators. All forward-looking statements contained in this press release speak only as of the date on which they were made. Acasti undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as required by applicable securities laws. For more information, please contact: Acasti Contact: Prashant KohliChief Executive OfficerTel: 450-686-4555Email:info@acastipharma.comwww.acasti.com Investor Relations: LifeSci AdvisorsMike MoyerManaging DirectorPhone: 617-308-4306Email: mmoyer@lifesciadvisors.com —tables to follow— ACASTI PHARMA INC.Consolidated Balance Sheets March 31, 2024 March 31, 2023 (Expressed in thousands except share data) $ $ Assets Current assets: Cash and cash equivalents 23,005 27,875 Short-term investments — 15 Receivables 722 802 Prepaid expenses 283 598 Total current assets 24,010 29,290 Operating lease right of use asset — 463 Equipment, net 24 104 Intangible assets 41,128 41,128 Goodwill 8,138 8,138 Total assets 73,300 79,123 Liabilities and Shareholders’ equity Current liabilities: Trade and other payables 1,684 3,336 Operating lease liability — 75 Total current liabilities 1,684 3,411 Derivative warrant liabilities 4,359 — Operating lease liability — 410 Deferred tax liability 5,514 7,347 Total liabilities 11,557 11,168 Commitments and contingencies Shareholders’ equity: Class A common shares, no par value per share; unlimited shares authorized; 9,399,404 and 7,435,533 shares issued and outstanding as of March 31, 2024 and 2023,respectively 261,038 258,294 Class B, C, D and E common shares, no par value per share; unlimited shares authorized; none issued and outstanding — — Additional paid-in capital 17,862 13,965 Accumulated other comprehensive loss (6,038) (6,038)Accumulated deficit (211,119) (198,266)Total shareholders’ equity 61,743 67,955 Total liabilities and shareholders’ equity 73,300 79,123 ACASTI PHARMA INC.Consolidated Statements of Operations and Comprehensive Loss Year ended March 31, 2024 Year ended March 31, 2023 (Expressed in thousands, except share and per data) $ $ Operating expenses Research and development expenses, net of government assistance (4,683) (9,972)General and administrative expenses (6,432) (7,614)Sales and marketing (252) (661)Restructuring cost (1,485) — Impairment of intangible assets — (28,682)Impairment of goodwill — (4,826)Impairment of assets held for sale — (400)Loss from operating activities (12,852) (52,155) Foreign exchange loss (16) (72)Change in fair value of derivative warrant liabilities (2,728) 10 Interest income and other expense, net 911 246 Total other income (expense), net (1,833) 184 Loss before income tax benefit (14,685) (51,971) Income tax benefit 1,832 9,542 Net loss and total comprehensive loss (12,853) (42,429) Basic and diluted loss per share (1.35) (5.71) Weighted average number of shares outstanding 9,529,123 7,435,472
SHL Telemedicine Confirms Discussions Regarding a Possible Investment in its Israeli Activity
June 20, 2024 06:32 AM Eastern Daylight Time TEL AVIV & ZURICH & NEW YORK–(BUSINESS WIRE)–SHL Telemedicine Ltd. (NASDAQ: SHLT, SIX: SHLTN;) (“SHL” or the “Company”), a leading provider and developer of advanced personal telemedicine solutions, has clarified today that it is in discussions with Discount Capital Ltd. the investment […]
GE HealthCare and MediView Announce the World’s First Installation and Clinical Use of Augmented Reality Interventional Suite that Aims to Transform the Practice of Interventional Radiology
June 20, 2024 10:47 AM Eastern Daylight Time CHICAGO–(BUSINESS WIRE)–Today GE HealthCare (Nasdaq: GEHC), a leading global medical technology, pharmaceutical diagnostics, and digital solutions innovator, and MediView XR Inc., a leading clinical augmented reality med-tech company, announced the successful first installation and clinical cases using the OmnifyXR Interventional Suite1 at North […]
Silence Therapeutics Announces Positive Topline 48-Week Data from Phase 2 Study of Zerlasiran in Patients with Elevated Lipoprotein(a)
LONDON–(BUSINESS WIRE)–Silence Therapeutics plc, Nasdaq: SLN (“Silence” or the “Company”), an experienced and innovative biotechnology company committed to transforming people’s lives by silencing diseases through precision engineered medicines, today announced positive topline 48-week data from the ALPACAR-360 phase 2 study of zerlasiran (SLN360) in 178 subjects with baseline lipoprotein(a), or […]
Vizient examines pulsed field ablation, wearables for women’s cardiovascular health in latest Medical Device Tech Watch
IRVING, Texas–(BUSINESS WIRE)–Vizient, Inc. examines pulsed field ablation technologies for atrial fibrillation and the role wearable monitoring devices can play in women’s cardiovascular health in the latest Medical Device Tech Watch. “By working closely with providers, we have developed best practices for operationalizing the use of these innovative technologies and tools.” Post […]
Microbot Medical Partners with Brigham and Women’s Hospital for Its Pivotal Human Clinical Trial
Following FDA Approval to Commence the Clinical Trial, an Official Site Initiation Has Taken Place as Preparation for Patient Enrollment Advances Multiple Robotic Systems Already Received by the Site to Allow Inventory Readiness in Support of Trial BRAINTREE, Mass., June 20, 2024 (GLOBE NEWSWIRE) — Microbot Medical Inc. (Nasdaq: MBOT) announced its agreement with Brigham and Women’s Hospital (BWH), a leading academic medical center located in Boston, Massachusetts, to serve as one of the sites to perform the pivotal human clinical trial for its LIBERTY® Endovascular Robotic Surgical System, as part of its Investigational Device Exemption (“IDE”) application. This development, previously announced on June 17, 2024, follows the U.S. Food and Drug Administration’s approval to commence Microbot’s pivotal human clinical trial. The Company has completed the Site Initiation Visit, during which BWH clinical staff was trained on the clinical study protocols and the use of the LIBERTY® Endovascular Robotic Surgical System. In addition, the first shipment of LIBERTY investigational systems arrived at BWH this week in support of the clinical trial. Dr. Dmitry Rabkin, MD, PhD (Assistant Chief, Division of Angiography & Interventional Radiology), will lead the study for the site as principal investigator at BWH. “We are pleased to work with Dr. Rabkin and the team at Brigham and Women’s Hospital on this clinical study,” commented Harel Gadot, CEO, President and Chairman of Microbot Medical. “We believe their commitment to research and the advancement of science make them an ideal clinical study site.” The Company is in the process of engaging additional leading centers to participate in the clinical trial. About Microbot Medical Microbot Medical Inc. (NASDAQ: MBOT) is a clinical stage medical device company that specializes in transformational micro-robotic technologies, with the goals of improving clinical outcomes for patients and increasing accessibility through the natural and artificial lumens within the human body. The Investigational LIBERTY® Endovascular Robotic Surgical System aims to improve the way surgical robotics are being used in endovascular procedures today, by eliminating the need for large, cumbersome, and expensive capital equipment, while reducing radiation exposure and physician strain. The Company believes the LIBERTY® Endovascular Robotic Surgical System’s remote operation has the potential to be the first system to democratize endovascular interventional procedures. Further information about Microbot Medical is available at http://www.microbotmedical.com. Safe Harbor Statements to future financial and/or operating results, future growth in research, technology, clinical development, and potential opportunities for Microbot Medical Inc. and its subsidiaries, along with other statements about the future expectations, beliefs, goals, plans, or prospects expressed by management, constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and the Federal securities laws. Any statements that are not historical fact (including, but not limited to statements that contain words such as “will,” “believes,” “plans,” “anticipates,” “expects” and “estimates”) should also be considered to be forward-looking statements. Forward-looking statements involve risks and uncertainties, including, without limitation, market conditions, risks inherent in the development and/or commercialization of the LIBERTY® Endovascular Robotic Surgical System, the outcome of its studies to evaluate the LIBERTY® Endovascular Robotic Surgical System, uncertainty in the results of pre-clinical and clinical trials or regulatory pathways and regulatory approvals, including whether the Company’s pivotal study in humans is successful, any failure or inability to recruit physicians and clinicians to serve as primary investigators to conduct regulatory studies which could adversely affect or delay such studies, disruptions resulting from new and ongoing hostilities between Israel and the Palestinians and other neighboring countries, any lingering uncertainty resulting from the COVID-19 pandemic, need and ability to obtain future capital, and maintenance of intellectual property rights. Additional information on risks facing Microbot Medical can be found under the heading “Risk Factors” in Microbot Medical’s periodic reports filed with the Securities and Exchange Commission (SEC), which are available on the SEC’s web site at www.sec.gov. Microbot Medical disclaims any intent or obligation to update these forward-looking statements, except as required by law. Investor Contact:Michal EfratyIR@microbotmedical.com