Clinical investigators complete record enrollment in real-world Registry, HEAL (An All-comers Registry of the MicroStent PeripHeral Vascular StEnt in subjects with PeripherAl ArteriaL Disease)
WILMINGTON, Mass., June 18, 2024 /PRNewswire/ — Micro Medical Solutions — Micro Medical Solutions, a leader in providing innovative medical device solutions, is excited to announce that it has achieved a major milestone with the enrollment of its 200th patient in the HEAL Registry. This unique registry is focused on gathering real world evidence using its MicroStent technology for treatment of patients with Chronic Limb-Threatening Ischemia (CLTI) and Critical Limb Ischemia (CLI).
CLTI and CLI are debilitating conditions that affect the blood vessels in the legs and feet. Those afflicted with this disease often suffer from severe pain, numbness, and open non-healing wounds. Without intervention, these conditions can lead to amputation, loss of mobility, and death. Micro Medical Solutions is committed to providing a potential solution for this medically complex and underserved patient population.
The HEAL Registry, enrolling patients at ten sites in EU countries, including Italy, Germany, Belgium, Austria and the Netherlands, is crucial clinical research in evaluating the use of MMS’ MicroStent to treat real world patients afflicted with CLI/CLTI. HEAL’s research endpoints provide real world evidence of the effectiveness and safety of treatment with the Micro Stent. With enrollment of 200 patients, the registry has reached a significant clinical and research milestone. The registry is also collecting specific data outcomes in below-the-ankle treatment, specific to wound healing. This data will be reported alongside MMS’ US IDE study data to the FDA in its premarket approval application (PMA).
“We are proud to have created the largest real-world cohort of patients and data collection for an implanted stent below the knee in the HEAL Registry,” said Greg Sullivan, CEO of Micro Medical Solutions. “This milestone is a testament to our commitment to providing innovative solutions for those suffering from CLI/CLTI. We are optimistic about the encouraging data that has emerged from the registry and look forward to utilizing this information to continue providing effective treatment options.”
Micro Medical Solutions completed enrollment in its STAND IDE study, a Randomized Controlled Trial, in the US in December of 2023. MMS expects to share this data in early 2025. STAND reflects controlled patient selection with required follow up visits, HEAL reflects general CLI/CLTI patient population treated in everyday practice while including required follow-up. The combination of RCT and RWE data will provide physicians with the opportunity and confidence to make treatment decisions using evidence-based medicine.
MMS is dedicated to making a positive impact on the lives of those affected by this limb threatening disease and is committed to using real world data to support its mission to support physicians in their management of these patients.
Thank you to the physicians and patients for their continued support as we strive towards providing effective and innovative solutions for those in need.
About Micro Medical Solutions
For more about Micro Medical Solutions, visit www.micromedicalsolutions.net.
SOURCE Micro Medical Solutions
Other News
Longeveron Raises $4.4 Million in Gross Proceeds from Warrant Exercise Transaction
MIAMI, June 18, 2024 (GLOBE NEWSWIRE) — Longeveron Inc. (NASDAQ: LGVN) (“Longeveron” or the “Company”), a clinical stage regenerative medicine biotechnology company developing cellular therapies for rare, life-threatening and chronic aging-related conditions, today announced the closing of its previously announced exercise of certain existing warrants to purchase an aggregate of 1,697,891 shares of its Class A common stock having an exercise price of $2.35 per share, originally issued in April 2024. The resale of the shares of Class A common stock issuable upon exercise of the existing warrants is registered pursuant to an effective registration statement on Form S-1 (File No. 333-278995). The gross proceeds to the Company from the exercise of the existing warrants were approximately $4.4 million, prior to deducting placement agent fees and estimated offering expenses payable by the Company. H.C. Wainwright & Co. acted as the exclusive placement agent for the offering. In consideration for the immediate exercise of the existing warrants for cash and the payment of $0.125 per new warrant, the Company issued new unregistered warrants to purchase up to an aggregate of 3,395,782 shares of Class A common stock. The new warrants are immediately exercisable at an exercise price of $2.50 per share and have a term of twenty-four months from the date of issuance. The Company intends to use the net proceeds from the transaction for its ongoing clinical and regulatory development of Lomecel-B™ for the treatment of several disease states and indications, including HLHS and Alzheimer’s disease, obtaining regulatory approvals, capital expenditures, working capital and other general corporate purposes. The new warrants described above were offered in a private placement under Section 4(a)(2) of the Securities Act of 1933, as amended (the “Securities Act”), and Regulation D promulgated thereunder and, along with the shares of Class A common stock issuable upon exercise of the new warrants, have not been registered under the Securities Act, or applicable state securities laws. Accordingly, the new warrants issued in the private placement and the shares of Class A common stock underlying the new warrants may not be offered or sold in the United States except pursuant to an effective registration statement or an applicable exemption from the registration requirements of the Securities Act and such applicable state securities laws. The Company has agreed to file a registration statement with the Securities and Exchange Commission covering the resale of the shares of Class A common stock issuable upon the exercise of the new warrants. This press release does not constitute an offer to sell or a solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any state or other jurisdiction in which such offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such state or other jurisdiction. About Longeveron Inc. Longeveron is a clinical stage biotechnology company developing regenerative medicines to address unmet medical needs. The Company’s lead investigational product is Lomecel-B™, an allogeneic medicinal signaling cell (MSC) therapy product isolated from the bone marrow of young, healthy adult donors. Lomecel-B™ has multiple potential mechanisms of action encompassing pro-vascular, pro-regenerative, anti-inflammatory, and tissue repair and healing effects with broad potential applications across a spectrum of disease areas. Longeveron is currently pursuing three pipeline indications: hypoplastic left heart syndrome (HLHS), Alzheimer’s disease, and Aging-related Frailty. For more information, visit www.longeveron.com or follow Longeveron on LinkedIn, X, and Instagram. Forward-Looking Statements Certain statements in this press release that are not historical facts are forward-looking statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, which reflect management’s current expectations, assumptions, and estimates of future operations, performance and economic conditions, and involve risks and uncertainties that could cause actual results to differ materially from those anticipated by the statements made herein. Forward-looking statements are generally identifiable by the use of forward-looking terminology such as “believe,” “expects,” “may,” “looks to,” “will,” “should,” “plan,” “intend,” “on condition,” “target,” “see,” “potential,” “estimates,” “preliminary,” or “anticipates” or the negative thereof or comparable terminology, or by discussion of strategy or goals or other future events, circumstances, or effects and include, but are not limited to, the potential for Lomecel-B™ to be a beneficial treatment for patients with HLHS and include, but are not limited to, the anticipated use of proceeds from the private placement. Factors that could cause actual results to differ materially from those expressed or implied in any forward-looking statements in this release include, but are not limited to, market and other conditions, adverse global conditions, including macroeconomic uncertainty; inability to raise additional capital necessary to continue as a going concern; our history of losses and inability to achieve profitability going forward; the absence of FDA-approved allogenic, cell-based therapies for HLHS or other cardiac-related indications; ethical and other concerns surrounding the use of stem cell therapy or human tissue; our exposure to product liability claims arising from the use of our product candidates or future products in individuals, for which we may not be able to obtain adequate product liability insurance; the adequacy of our trade secret and patent position to protect our product candidates and their uses: others could compete against us more directly, which could harm our business and have a material adverse effect on our business, financial condition, and results of operations; if certain license agreements are terminated, our ability to continue clinical trials and commercially market products could be adversely affected; the inability to protect the confidentiality of our proprietary information, trade secrets, and know-how; third-party claims of intellectual property infringement may prevent or delay our product development efforts; the inability to successfully develop and commercialize our product candidates and obtain the necessary regulatory approvals; we cannot market and sell our product candidates in the U.S. or in other countries if we fail to obtain the necessary regulatory approvals; final marketing approval of our product candidates by the FDA or other regulatory authorities for commercial use may be delayed, limited, or denied, any of which could adversely affect our ability to generate operating revenues; we may not be able to secure and maintain research institutions to conduct our clinical trials; ongoing healthcare legislative and regulatory reform measures may have a material adverse effect on our business and results of operations; if we receive regulatory approval of Lomecel-B™ or any of our other product candidates, we will be subject to ongoing regulatory requirements and continued regulatory review, which may result in significant additional expense; being subject to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with our therapeutic candidates; reliance on third parties to conduct certain aspects of our preclinical studies and clinical trials; interim, “topline” and preliminary data from our clinical trials that we announce or publish from time to time may change as more data become available and are subject to audit and verification procedures that could result in material changes in the final data; provisions in our certificate of incorporation and bylaws and Delaware law might discourage, delay or prevent a change in control of our company or changes in our management and, therefore, depress the market price of our Class A common stock; we have never commercialized a product candidate before and may lack the necessary expertise, personnel and resources to successfully commercialize any products on our own or together with suitable collaborators; and in order to successfully implement our plans and strategies, we will need to grow our organization, and we may experience difficulties in managing this growth. Further information relating to factors that may impact the Company’s results and forward-looking statements are disclosed in the Company’s filings with the Securities and Exchange Commission, including Longeveron’s Annual Report on Form 10-K for the year ended December 31, 2023, filed with the Securities and Exchange Commission on February 27, 2024, as amended by the Annual Report on Form 10-K/A filed March 11, 2024, its Quarterly Reports on Form 10-Q, and its Current Reports on Form 8-K. The forward-looking statements contained in this press release are made as of the date of this press release, and the Company disclaims any intention or obligation, other than imposed by law, to update or revise any forward-looking statements, whether as a result of new information, future events, or otherwise. Investor ContactDerek ColeInvestor Relations Advisory Solutionsderek.cole@iradvisory.com
Haemonetics Launches Limited Market Release for New VASCADE MVP XL Vascular Closure Device
BOSTON, June 18, 2024 /PRNewswire/ — Haemonetics Corporation (NYSE: HAE), a global medical technology company focused on delivering innovative solutions to drive better patient outcomes, has launched a limited market release of its new VASCADE MVP® XL mid-bore venous closure device. The VASCADE MVP XL system expands Haemonetics’ VASCADE® portfolio of vascular closure systems featuring an innovative collapsible disc technology and a proprietary resorbable collagen patch designed to promote rapid hemostasis.
Haemonetics’ current VASCADE portfolio includes the VASCADE system, designed for “small-bore” femoral arterial and venous closure with standard 5-6/7F procedural sheaths, and the VASCADE MVP® system, designed for “mid-bore” multi-access femoral venous closure with 6-12F procedural sheaths. The upsized VASCADE MVP XL system utilizes 58% more collagen and a larger disc than the current VASCADE MVP system, providing a robust closure solution for procedures requiring 10-12F sheaths (up to 15F in outer diameter) such as cryoablation and left atrial appendage closure for atrial fibrillation patients.
“With VASCADE MVP XL, Haemonetics continues to expand its presence and broaden its reach in the $2.7 billion total addressable market for vascular closure solutions,” said Stew Strong, President of Global Hospital at Haemonetics. “The introduction of VASCADE MVP XL underscores our commitment to innovation and improving patient care, as we enhance our range of vascular closure solutions to address increasing demand for catheter-based ablation technologies. We are enthusiastic about the initial launch of VASCADE MVP XL and anticipate a full market release later this year.”
The VASCADE MVP XL system earned pre-market approval from the U.S. Food and Drug Administration this spring. The limited market release follows the first procedure performed using VASCADE MVP XL by Dr. Tom McElderry, Section Chief, Electrophysiology and Co-Director Heart & Vascular Center at the University of Alabama at Birmingham.
About Haemonetics
Haemonetics (NYSE: HAE) is a global healthcare company dedicated to providing a suite of innovative medical products and solutions for customers, to help them improve patient care and reduce the cost of healthcare. Our technology addresses important medical markets: blood and plasma component collection, the surgical suite and hospital transfusion services. Haemonetics’ Global Hospital business provides a range of solutions to address the needs of hospitals, including Interventional Technologies for electrophysiology and interventional cardiology, and Blood Management Technologies that include diagnostics to help inform treatment decisions, technologies to help avoid unnecessary allogeneic transfusions and solutions to help optimize management of blood products. To learn more about Haemonetics, visit www.haemonetics.com.
Cautionary Statement Regarding Forward-Looking Information
Any statements contained in this press release that do not describe historical facts may constitute forward-looking statements. Forward-looking statements in this press release may include, without limitation, statements regarding plans and objectives of management for the operation of Haemonetics, including statements regarding potential benefits associated with the Vascade MVP XL vascular closure device and Haemonetics’ plans or objectives related to the commercialization of such product. Such forward-looking statements are not meant to predict or guarantee actual results, performance, events or circumstances and may not be realized because they are based upon Haemonetics’ current projections, plans, objectives, beliefs, expectations, estimates and assumptions and are subject to a number of risks and uncertainties and other influences. Actual results and the timing of certain events and circumstances may differ materially from those described by the forward-looking statements as a result of these risks and uncertainties. Factors that may influence or contribute to the inaccuracy of the forward-looking statements or cause actual results to differ materially from expected or desired results may include, without limitation, product quality; market acceptance; the effect of global economic and political conditions; and the impact of competitive products and pricing. These and other factors are identified and described in more detail in Haemonetics’ periodic reports and other filings with the U.S. Securities and Exchange Commission. Haemonetics does not undertake to update these forward-looking statements.
Investor Contacts:
Olga Guyette, Vice President-Investor Relations & Treasury
David Trenk, Manager-Investor Relations
(781) 356-9763
(203) 733-4987
[email protected]
[email protected]
Media Contact:
Josh Gitelson, Senior Director-Global Communications
(781) 356-9776
[email protected]
SOURCE Haemonetics Corporation
Morton Plant Hospital First in Tampa Bay to Use New Treatment for Restenosis
CLEARWATER, Fla., June 18, 2024 /PRNewswire/ — Morton Plant Hospital recently became the first hospital in the Tampa Bay area to use a drug-coated balloon to treat in-stent restenosis. The hospital was ranked #1 in Florida for Cardiac Surgery in 2023 by Healthgrades, where the area’s first robotic-assisted coronary artery bypass surgery took place in early June. BayCare Medical Group’s Interventional Cardiologist Lang Lin, MD, and her expert team of medical professionals at Morton Plant Hospital performed the first procedure with the drug-coated balloon earlier this month.
Continue Reading
Cardiologist Lang Lin, MD, and her team from Morton Plant Hospital’s cardiac catheterization lab celebrate using a new tool to treat restenosis in Clearwater, Fla.
Restenosis occurs when an artery that has previously been stented narrows again because of plaque or scar tissue.
Treating in-stent restenosis remains a significant challenge for both patients and cardiologists, even 20 years after drug-eluting stents (which are coated with medicine to keep arteries open) became the standard of care.” Dr. Lin said. Until now, in-stent restenosis has most commonly been treated in one of two ways: by placing an additional stent in the stent of the artery that has narrowed again or by performing coronary artery bypass surgery. The challenge with two or more layers of stents is that additional stents become less effective and more difficult to treat. Coronary artery bypass surgery has increased risks with older patients and sometimes limited bypass target vessels. The new drug-coated balloon, AGENT™ from Boston Scientific, was approved by the U.S. Food and Drug Administration (FDA) for commercial use in March. The balloon catheter, the first drug-coated coronary balloon in the United States, reopens the narrowed stent and releases the high concentration of drugs to prevent scar tissue from forming and blocking the artery wall again. “We are pleased to have the opportunity to offer this cutting-edge treatment to our patients,” Dr. Lin said. “The new technology will help us to treat certain patients with in-stent restenosis without the need for another stent, invasive surgery or brachytherapy (radiation).” For more information on heart and vascular services at BayCare: BayCare’s Heart and Vascular Services. About Morton Plant HospitalSince 1916, Morton Plant Hospital has been committed to improving the health of all it serves through community-owned health care services that set the standard for high-quality, compassionate care. The 599-bed hospital is proud to offer nationally recognized care delivered in more than 50 specialty areas. Part of BayCare Health System, Morton Plant Hospital offers innovative, accessible and quality services to provide our community with a lifetime of compassionate, convenient care. The hospital is located at 300 Pinellas Street in Clearwater, Florida. For more information, visit BayCare.org/MPH.About BayCareBayCare is a leading not-for-profit health care system that connects individuals and families to a wide range of services at 16 hospitals and hundreds of other convenient locations throughout the Tampa Bay and central Florida regions. The system is West Central Florida’s largest provider of behavioral health and pediatric services and its provider group, BayCare Medical Group, is one of the largest in the region. BayCare’s diverse network of ambulatory services includes laboratories, imaging, surgical centers, BayCare Urgent Care locations, wellness centers and one of Florida’s largest home care agencies, BayCare HomeCare. BayCare’s mission is to improve the health of all it serves through community-owned, health care services that set the standard for high-quality, compassionate care. For more information visit BayCare.org.SOURCE BayCare Health System
Merit Medical Systems to Announce Second Quarter 2024 Results on August 1, 2024
SOUTH JORDAN, Utah, June 18, 2024 (GLOBE NEWSWIRE) — Merit Medical Systems, Inc. (NASDAQ: MMSI), a leading global manufacturer and marketer of healthcare technology, announced today that it will release its financial results for the quarter ended June 30, 2024, after the close of the stock market on Thursday, August 1, 2024. Merit will hold its investor conference call on the same day (Thursday, August 1, 2024) at 5:00 p.m. Eastern (4:00 p.m. Central, 3:00 p.m. Mountain, and 2:00 p.m. Pacific). To access the conference call, please pre-register using the following link. Registrants will receive confirmation with dial-in details. A live webcast and slide deck can be accessed using this link. A link to both register for the conference call and view the webcast will be made available at merit.com. ABOUT MERIT Founded in 1987, Merit Medical Systems, Inc. is engaged in the development, manufacture, and distribution of proprietary disposable medical devices used in interventional, diagnostic, and therapeutic procedures, particularly in cardiology, radiology, oncology, critical care, and endoscopy. Merit serves client hospitals worldwide with a domestic and international sales force and clinical support team totaling more than 700 individuals. Merit employs approximately 7,000 people worldwide.
RADPAIR and NewVue Announce Groundbreaking Partnership to Enhance Radiologist Wellbeing and Workflow Efficiency
TAMPA, Fla. and KNOXVILLE, Tenn., June 18, 2024 /PRNewswire/ — RADPAIR and NewVue are excited to announce a strategic partnership that will be the first of its kind in the radiology industry. This collaboration integrates RADPAIR’s advanced AI diagnostic reporting capabilities into NewVue’s workflow orchestrator, creating the industry’s first cloud-native solution to enhance radiologists’ well-being and job satisfaction while improving workflow and report quality.
The combined solutions will transform how radiologists and practices interact with their worklists, manage clinical information, dictate reports, follow up on cases, and track productivity. Leveraging modern capabilities like AI and large language models (LLMs), this next-generation cloud product delivers an innovative, efficient, and enjoyable experience.
EmpowerSuite automatically generates a curated worklist based on the radiologist’s profile, considering their preferred specialties, credentialing, insurance coverage, scheduled shifts, and physical location. This next-generation approach to workload distribution and management departs from traditional first-generation worklists that require extensive maintenance of rules and lists. Radiologists can also adjust the reading pace and cadence based on their mood, improving job satisfaction.
Studies opened from the worklist launch PACS and the EmpowerSuite Clinical information screen. This “radiologist cockpit” uses AI to summarize all available prior reports and clinical information, saving radiologists time by eliminating the need to search multiple systems. The cockpit also hosts the RADPAIR AI-powered reporting system, which generates reports from conversational inputs. Automatic reporting and dynamic editing features allow for near real-time report generation and simple click-and-drag editing for minor revisions. The cloud-native platform ensures easy sign-in with zero context switching, allowing radiologists to focus entirely on diagnostics. This innovative workflow and reporting system integrates with existing PACS and replaces legacy VR and dictation systems.
“We’ve been really thinking about how to make the lives of radiologists fun again while increasing quality. Our new cutting-edge classification system PAIR Insights allows radiologists to seamlessly insert guidelines into reports, enhancing understanding for both clinicians and patients. This partnership with NewVue underscores our commitment to improving the radiology experience,” said Dr. Avez Rizvi, Founder & CEO of RADPAIR.
“We are excited to join forces with RADPAIR to create a solution that enhances the efficiency of radiology reporting and significantly improves the well-being of radiologists. By integrating RADPAIR’s cutting-edge technology into our workflow orchestrator, we are setting a new standard for radiology practices,” said Kyle Lawton, CEO of NewVue.
###
About RADPAIR:RADPAIR is a leader in innovative radiology solutions, dedicated to improving the radiologist’s experience and the quality of patient care. With a focus on smart systems and trusted guidelines, RADPAIR sets the standard in radiology reporting.
About NewVue:NewVue specializes in workflow orchestration for healthcare providers, delivering solutions that enhance operational efficiency and care quality. NewVue’s platform integrates seamlessly with existing systems to optimize workflows and improve patient outcomes.
Media ContactFatima BaigRADPAIR310-766-8911[email protected]
SOURCE RADPAIR
Analysis of INPEFA® (Sotagliflozin) Cost-Effectiveness Published in JACC: Heart Failure, the Peer-Reviewed Journal of the American College of Cardiology
New analysis of the pivotal Phase 3 SOLOIST-WHF trial demonstrates INPEFA cost-effectiveness Findings consistent with another study recently published in the Journal of Comparative Effectiveness Research THE WOODLANDS, Texas, June 18, 2024 (GLOBE NEWSWIRE) — Lexicon Pharmaceuticals, Inc. (Nasdaq: LXRX) today announced that the peer-reviewed Journal of the American College of Cardiology: Heart Failure has published a research paper concluding that INPEFA® (sotagliflozin) is cost-effective for people with diabetes and recent worsening heart failure using commonly accepted willingness-to-pay thresholds. “Our research team believes that this study is an important contribution to the economic evaluation of sotagliflozin, a novel SGLT inhibitor, from the perspective of the U.S. healthcare system. Our results demonstrated that in people with diabetes and recent worsening heart failure, sotagliflozin is cost-effective at commonly accepted willingness-to-pay thresholds,” said William S. Weintraub, MD, MACC, FAHA, FESC, director of Population Health Research at MedStar Health Research Institute, and the lead author of the research paper. The analysis was conducted from a U.S. healthcare sector perspective, in accordance with Consolidated Health Economic Evaluation Reporting Standards. Results from this study showed that lifetime quality-adjusted life-years (QALYs) were 4.43 and 4.04 in the INPEFA and placebo groups, respectively. In another study, “Cost-effectiveness of sotagliflozin for the treatment of patients with diabetes and recent worsening heart failure,” recently published in the Journal of Comparative Effectiveness Research, the research team used published real-world data to derive baseline event frequencies and SOLOIST-WHF study data to estimate the efficacy of INPEFA. According to the research results, the use of INPEFA led to a net gain in QALYs of 0.425 for INPEFA versus standard of care. The investigators concluded that INPEFA is a cost-effective addition to standard of care for patients hospitalized with heart failure and comorbid diabetes. “The data published in these two peer-reviewed journals reinforce our position that in addition to providing meaningful clinical benefits to heart failure patients, INPEFA provides significant financial value for payors and the U.S. healthcare system,” said Craig Granowitz, M.D., Ph.D., Lexicon’s senior vice president and chief medical officer. The Journal of the American College of Cardiology: Heart Failure manuscript can be accessed here. About Lexicon Pharmaceuticals Lexicon is a biopharmaceutical company with a mission of pioneering medicines that transform patients’ lives. Through the Genome5000™ program, Lexicon’s unique genomics target discovery platform, Lexicon scientists studied the role and function of nearly 5,000 genes and identified more than 100 protein targets with significant therapeutic potential in a range of diseases. Through the precise targeting of these proteins, Lexicon is pioneering the discovery and development of innovative medicines to treat disease safely and effectively. Lexicon has commercially launched one of these medicines, INPEFA® (sotagliflozin) in the United States, and has a pipeline of other promising drug candidates in discovery and clinical and preclinical development in neuropathic pain, diabetes and metabolism and other indications. For additional information, please visit www.lexpharma.com. About INPEFA® (sotagliflozin) Discovered using Lexicon’s unique approach to gene science, INPEFA® (sotagliflozin) is an oral inhibitor of two proteins responsible for glucose regulation known as sodium-glucose cotransporter types 2 and 1 (SGLT2 and SGLT1). SGLT2 is responsible for glucose and sodium reabsorption by the kidney and SGLT1 is responsible for glucose and sodium absorption in the gastrointestinal tract. Sotagliflozin has been studied in multiple patient populations encompassing heart failure, diabetes, and chronic kidney disease in clinical studies involving approximately 20,000 patients. INDICATION INPEFA is indicated to reduce the risk of cardiovascular death, hospitalization for heart failure, and urgent heart failure visit in adults with: heart failure or type 2 diabetes mellitus, chronic kidney disease, and other cardiovascular risk factors IMPORTANT SAFETY INFORMATION Dosing: Assess renal function and volume status and, if necessary, correct volume depletion prior to initiation of INPEFA. INPEFA dosing for patients with decompensated heart failure may begin when patients are hemodynamically stable, including when hospitalized or immediately upon discharge. Contraindications: INPEFA is contraindicated in patients with hypersensitivity to INPEFA or any of its components. Ketoacidosis: INPEFA increases the risk of ketoacidosis in patients with type 1 diabetes mellitus (T1DM). Type 2 diabetes Mellitus (T2DM) and pancreatic disorders are also risk factors. The risk of ketoacidosis may be greater with higher doses. There have been postmarketing reports of fatal events of ketoacidosis in patients with type 2 diabetes using sodium glucose transporter 2 (SGLT2) inhibitors. Before initiating INPEFA, assess risk factors for ketoacidosis. Consider ketone monitoring in patients with T1DM and consider ketone monitoring in others at risk for ketoacidosis and educate patients on the signs/symptoms of ketoacidosis. Patients receiving INPEFA may require monitoring and temporary discontinuation of therapy in clinical situations known to predispose to ketoacidosis. INPEFA is not indicated for glycemic control. Assess patients who present with signs and symptoms of metabolic acidosis or ketoacidosis, regardless of blood glucose level. If suspected, discontinue INPEFA, evaluate, and treat promptly. Monitor patients for resolution of ketoacidosis before restarting INPEFA. Volume Depletion: INPEFA can cause intravascular volume depletion which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine. There have been post-marketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors. Patients with impaired renal function (eGFR < 60 mL/min/1.73 m2), elderly patients, or patients on loop diuretics may be at increased risk for volume depletion or hypotension. Before initiating INPEFA in patients with one or more of these characteristics, assess volume status and renal function, and monitor for signs and symptoms of hypotension during therapy. Urosepsis and Pyelonephritis: Treatment with SGLT2 inhibitors, including INPEFA, increases the risk for urinary tract infections. Serious urinary tract infections including urosepsis and pyelonephritis requiring hospitalization have been reported. Evaluate patients for signs and symptoms of urinary tract infections and treat promptly. Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues: Insulin and insulin secretagogues are known to cause hypoglycemia. INPEFA may increase the risk of hypoglycemia when combined with insulin or an insulin secretagogue. Therefore, a lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used with INPEFA. Necrotizing Fasciitis of the Perineum (Fournier’s Gangrene): Reports of Fournier’s Gangrene, a rare but serious and life-threatening necrotizing infection requiring urgent surgical intervention, have been identified in post-marketing surveillance in patients with diabetes mellitus receiving SGLT2 inhibitors. Assess patients who present with pain, tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise. If suspected, start treatment immediately with broad-spectrum antibiotics and, if necessary, surgical debridement. Discontinue INPEFA, closely monitor patient signs and symptoms, and provide appropriate alternative therapy for heart failure. Genital Mycotic Infections: INPEFA increases the risk of genital mycotic infections. Monitor and treat as appropriate. Urinary Glucose Test and 1,5-anhydroglucitol (1,5-AG) Assay: these are not reliable for patients taking SGLT2 inhibitors. Use alternative testing methods to monitor glucose levels. Common Adverse Reactions: the most commonly reported adverse reactions (incidence ≥ 5%) were urinary tract infection, volume depletion, diarrhea, and hypoglycemia. Drug Interactions: Digoxin: Monitor patients appropriately as there is an increase in the exposure of digoxin when coadministered with INPEFA 400 mg.Uridine 5'-diphospho-glucuronosyltransferase (UGT) Inducer: The coadministration of rifampicin, an inducer of UGTs, with sotagliflozin resulted in a decrease in the exposure of sotagliflozin. Lithium: Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. Monitor serum lithium concentration more frequently during INPEFA initiation and with dosage changes. Use in Specific Populations: Pregnancy and Lactation: INPEFA is not recommended during the second and third trimesters of pregnancy, nor while breastfeeding. Geriatric Use: No INPEFA dosage change is recommended based on age. No overall differences in efficacy were detected between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. Elderly patients may be at increased risk for volume depletion adverse reactions, including hypotension. Renal Impairment: INPEFA was evaluated in patients with chronic kidney disease (eGFR 25 to 60 mL/min/1.73 m2) and in patients with heart failure with eGFR
Cordis Announces Positive 24-Month Results from Superficial Femoral Artery Study, Confirming Sustained Benefits of SELUTION SLR™ (sustained limus release) Drug-Eluting Balloon in Complex Patients
MIAMI LAKES, Fla., June 18, 2024 /PRNewswire/ — Cordis, a global leader in the development and manufacturing of interventional cardiovascular and endovascular technology, today announces positive 24-month results from the SELUTION SFA Japan Trial. The prospective, multi-center, single arm trial is designed to assess the safety and efficacy of SELUTION SLR™ Drug-Eluting Balloon (DEB) for the endovascular therapy of de novo and non-stented restenotic lesions in the superficial femoral artery (SFA) and the popliteal artery (PA). The findings were presented at the Japan Endovascular Treatment Conference (JET) 2024.
Continue Reading
“The SELUTION SLR™ DEB outcomes demonstrate patient benefits that are sustained out to 24 months and can match proven paclitaxel DCB performance in a complex patient population,” said Dr. Osamu Iida, Osaka Police Hospital Cardiovascular Division, Osaka, Japan. “These are promising results that build confidence in Limus drug-eluting balloons being a safe and effective option for SFA treatment while avoiding any paclitaxel related concerns.”
The long-term follow-up of SELUTION SLR™ DEB confirmed efficient sirolimus drug transfer and retention with safety and efficacy sustained through 24 months in a complex population that included 60.3% diabetics, mean lesion length of 127 mm, 17.2% total occlusions, and 47.8% involvement of the popliteal.
At 24 months, SELUTION SLR™ DEB delivered primary patency of 83.0% and CD-TLR of 95.4% achieving best in class efficacy results without compromise.
The SELUTION SFA Japan trial results demonstrate consistency of the SELUTION SLR™ DEB in being a safe and effective treatment option for femoropopliteal disease.
“The SELUTION SFA Japan trial continues to build confidence in the SELUTION SLR™ DEB technology. The SELUTION SLR™ DEB delivers some of the highest patency rates amongst SFA paclitaxel DCB studies and differentiates from other Limus-based devices. The ability to achieve durable clinical results in a challenging patient population provides clinicians a solution that will offer value to real world patients,” said George Adams, M.D. and Chief Medical Officer at Cordis. “We are excited for the evolution of patient care and to be leading that journey by expanding access to SELUTION SLR™ DEB.”Cordis is committed to generating coronary and peripheral clinical evidence that changes the standard of care. The SELUTION SFA Japan trial is one of the first of several large trials in the SELUTION SLR™ DEB clinical trial portfolio. Long term results validate previous clinical experience and builds confidence and excitement for data to come from four major randomized controlled trials and one real world registry data sets currently underway across coronary, superficial femoral artery and below the knee arteries.About SELUTION SLR ™ DEBSELUTION SLR™ DEB uses MicroReservoirs which contain a mixture of biodegradable polymer intermixed with the anti-restenotic drug sirolimus, applied as a coating on the surface of an angioplasty balloon. These MicroReservoirs are designed to provide controlled and sustained release of the drug. The proprietary CELL ADHERENT TECHNOLOGY (CAT)™ enables the MicroReservoirs to be coated onto balloons and efficiently transferred to adhere to the vessel lumen when delivered via expansion of the balloon.SELUTION SLR™ Drug-Eluting Balloon coronary, SFA, and BTK platforms are commercially available in Europe, the Middle East, and several markets across Asia and Latin America and most other countries where the CE mark is recognized.SELUTION SLR™ Drug-Eluting Balloon is investigational in the United States and not approved for sale or distribution.About CordisCordis is a global leader in the development and manufacturing of interventional cardiovascular and endovascular technologies. For over 60 years, we have remained committed to improving patients’ lives through innovation, enhanced clinical outcomes, education, and service. Our mission is to relentlessly pursue our dedication to transform cardiovascular care.For more information about Cordis, please visit www.cordis.com and follow Cordis on LinkedIn.Media Contact:Chris BinghamVice President, Global Market & Strategy[email protected]SOURCE Cordis
Boston Scientific Announces Agreement to Acquire Silk Road Medical, Inc.
Acquisition adds innovative technology for stroke prevention to vascular portfolio
MARLBOROUGH, Mass., June 18, 2024 /PRNewswire/ — Boston Scientific Corporation (NYSE: BSX) today announced it has entered into a definitive agreement to acquire Silk Road Medical, Inc. (Nasdaq: SILK), a medical device company that has developed an innovative platform of products to prevent stroke in patients with carotid artery disease through a minimally invasive procedure called transcarotid artery revascularization (TCAR). The purchase price is $27.50 per share, reflecting an enterprise value of approximately $1.16 billion.i
Carotid artery disease is the cause of one-third of all strokes and a condition in which the carotid arteries in the neck become narrowed or blocked due to the buildup of plaque.ii Treatment options for this disease include medical therapy management, placement of a stent, or surgery to reduce the risk of stroke. The TCAR procedure involves accessing the carotid artery through a small incision in the neck and temporarily reversing blood flow away from the brain to prevent plaque from dislodging and causing a stroke. A stent is then placed at the site of the blockage for long-term plaque stabilization and future stroke prevention.
“The TCAR platform developed by Silk Road Medical is a notable advancement in the field of vascular medicine, which has revolutionized stroke prevention and the treatment of carotid artery disease,” said Cat Jennings, president, Vascular, Peripheral Interventions, Boston Scientific. “We believe the addition of this clinically differentiated technology to our vascular portfolio demonstrates our continued commitment to provide meaningful innovation for physicians who care for patients with peripheral vascular disease.”
The TCAR system gained U.S. Food and Drug Administration approval in 2015 and is supported by several clinical studies demonstrating a reduced risk of stroke and other complications associated with traditional open surgery. The products sold by Silk Road Medical are the only devices commercially available for use during the TCAR procedure.
Boston Scientific expects to complete the transaction in the second half of 2024, subject to customary closing conditions. Silk Road Medical has guided to net revenue of approximately $194-198 million in 2024, representing 10-12% growth over the prior fiscal year.iii The impact to Boston Scientific adjusted earnings per share is expected to be immaterial in 2024 and 2025, and accretive thereafter. We expect the impact to GAAP earnings per share to be less accretive, or dilutive, as the case may be, due to amortization expense and acquisition-related net charges.
About Boston Scientific
Boston Scientific transforms lives through innovative medical technologies that improve the health of patients around the world. As a global medical technology leader for more than 40 years, we advance science for life by providing a broad range of high-performance solutions that address unmet patient needs and reduce the cost of health care. Our portfolio of devices and therapies helps physicians diagnose and treat complex cardiovascular, respiratory, digestive, oncological, neurological and urological diseases and conditions. Learn more at www.bostonscientific.com and connect on LinkedIn and X, formerly Twitter.
Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements may be identified by words like “anticipate,” “expect,” “project,” “believe,” “plan,” “estimate,” “intend” and similar words. These forward-looking statements are based on our beliefs, assumptions and estimates using information available to us at the time and are not intended to be guarantees of future events or performance. These forward-looking statements include, among other things, statements regarding financial and business impact of the transaction and anticipated benefits of the transaction, the closing of the transaction and the timing thereof, business plans and strategy, product launches and product performance and impact. If our underlying assumptions turn out to be incorrect, or if certain risks or uncertainties materialize, actual results could vary materially from the expectations and projections expressed or implied by our forward-looking statements. These factors, in some cases, have affected and in the future (together with other factors) could affect our ability to implement our business strategy and may cause actual results to differ materially from those contemplated by the forward-looking statements expressed in this press release. As a result, readers are cautioned not to place undue reliance on any of our forward-looking statements.
Risks and uncertainties that may cause such differences include, among other things: economic, political, competitive, reimbursement and regulatory conditions; geopolitical events; manufacturing, distribution and supply chain disruptions and cost increases; disruptions caused by cybersecurity events; disruptions caused by public health emergencies or extreme weather or other climate change-related events; labor shortages and increases in labor costs; variations in outcomes of ongoing and future clinical trials and market studies; new product introductions and the market acceptance of those products; market competition for our products; expected pricing environment; expected procedural volumes; demographic trends; the closing and integration of acquisitions, including our ability to achieve the anticipated benefits of the proposed transaction and successfully integrate Silk Road Medical’s operations; business disruptions (including disruptions in relationships with employees, customers and suppliers) following the announcement and/or closing of the proposed transaction; intellectual property rights; litigation; financial market conditions; the execution and effect of our business strategy, including our cost-savings and growth initiatives; future business decisions made by us and our competitors; the conditions to the completion of the proposed transaction, including receipt of the required regulatory approvals and clearances, may not be satisfied at all or in a timely manner; and the closing of the proposed transaction may not occur or may be delayed. New risks and uncertainties may arise from time to time and are difficult to predict. All of these factors are difficult or impossible to predict accurately and many of them are beyond our control. For a further list and description of these and other important risks and uncertainties that may affect our future operations, see Part I, Item 1A – Risk Factors in our most recent Annual Report on Form 10-K filed with the Securities and Exchange Commission (“SEC”), which we may update in Part II, Item 1A – Risk Factors in Quarterly Reports on Form 10-Q we have filed or will file hereafter. We disclaim any intention or obligation to publicly update or revise any forward-looking statements to reflect any change in our expectations or in events, conditions or circumstances on which those expectations may be based, or that may affect the likelihood that actual results will differ from those contained in the forward-looking statements, except as required by law. This cautionary statement is applicable to all forward-looking statements contained in this press release.
CONTACTS:Blake RouhaniMedia Relations(763) 494-2268[email protected]
Jon MonsonInvestor Relations(508) 683-5450[email protected]
i Enterprise value of approximately $1.16 billion based on approximately 46.0 million fully diluted shares (or approximately $1.26 billion for 100% of the equity) and $101 million net cash position as of March 31, 2024ii Weerd M Stroke 2010; Modus Health Group for 2022Vascularweb.org; Virani, Salim, et al. “Heart Disease and Stroke Statistics— 2020 Update.” American Heart Association, 3 Mar. 2020.iii https://investors.silkroadmed.com/news-releases/news-release-details/silk-road-medical-reports-first-quarter-2024-financial-results#:~:text=Silk%20Road%20Medical%20projects%20revenue,the%20Company’s%20prior%20year%20revenue.
SOURCE Boston Scientific Corporation
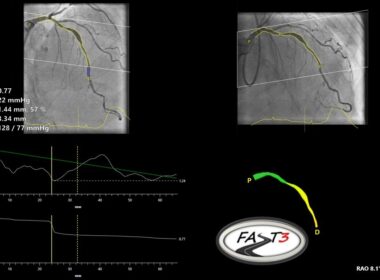
Pie Medical Imaging announces enrollment completion of the FASTIII clinical trial
MAASTRICHT, Netherlands, June 18, 2024 /PRNewswire/ — Pie Medical Imaging, a global leader in cardiac imaging, announced the completion of enrollment in FASTIII, a multi-center randomized clinical trial, which investigates the use of angiography-based vessel fractional flow reserve (CAAS vFFR) in patients undergoing coronary revascularization procedures. vFFR can assess whether a coronary artery narrowing is functionally significant and may require revascularization.
Continue Reading
CAAS vFFR Software
FASTIII is the largest non-inferiority trial running (having enrolled 2228 patients), in which an angiographically derived vFFR guided strategy is compared to a FFR guided strategy to guide coronary revascularization. The primary endpoint is a composite of all-cause death, any myocardial infarction, or any revascularization at 1-year post randomization.
Following the tremendous efforts of the principal investigator, Dr Joost Daemen (cardiologist at Thoraxcenter at the Erasmus University Medical Center, Rotterdam, The Netherlands), over 35 participating centers and ECRI (sponsor of the trial), the patient enrollment has concluded.
“The FASTIII trial is aimed to establish the role of vFFR to coronary revascularization in patients with intermediate coronary artery lesions. An important milestone was reached Friday, May 31st, 2024, when the last patient was enrolled”, said Joost Daemen Principal Investigator of the trial. “The next phase will consist of close follow-up of all patients who generously agreed to participate in the important trial. We hope to present our findings by the end of 2025″. Additionally,”PMI is committed to providing clinicians and patients with long-term coronary data to inform their treatment decisions, ” said René Guillaume, Managing Director at Pie Medical Imaging. “Our prior studies have shown diagnostic accuracy and reproducibility of vFFR calculation”. FASTIII will establish its role in routine clinical practice. The trial is funded by research grants from Pie Medical Imaging (Maastricht, The Netherlands) and Siemens Healthineers (Erlangen, Germany). The study is sponsored by ECRI (European Cardiovascular Research Institute, Rotterdam-the Netherlands). Cardialysis (Rotterdam, The Netherlands) is responsible for trial services including trial management and Core Laboratory activities.About Pie Medical ImagingPie Medical Imaging BV is a world leader in analysis and visualization of cardiovascular images. In Maastricht (The Netherlands), it hosts the global sales for the CAAS and 3mensio product lines.PMI and 3mensio Medical Imaging part of the Esaote Group, leader in the biomedical equipment sector. More information about PMI is available at www.piemedicalimaging.com Photo – https://mma.prnewswire.com/media/2438118/CAAS_vFFR.jpgLogo – https://mma.prnewswire.com/media/2438117/PMI_logo.jpg