BETHESDA, Md.–(BUSINESS WIRE)–CorVista Health, Inc., a leading digital health company dedicated to improving cardiovascular disease diagnosis announces expansion of their Medical Advisory Board with the appointment of Dr. Anjali Tiku Owens. “Dr. Tiku Owens’ wealth of experience and expertise in cardiology, particularly in genetic cardiomyopathy and novel therapies, will be […]
Other News
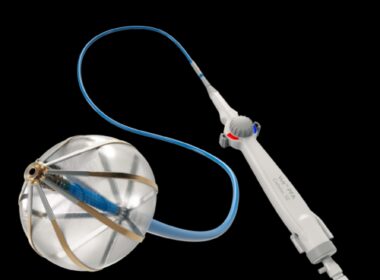
Pi-Cardia Receives FDA Breakthrough Device Designation for ShortCut™
REHOVOT, Israel–(BUSINESS WIRE)–Pi-Cardia Ltd., a global leader in the development of non-implant, catheter-based, leaflet modification solutions for treating heart valves, announced today it received Breakthrough Device Designation from the US Food and Drug Administration for ShortCut™ – the world’s first dedicated leaflet modification device facilitating valve-in-valve Transcatheter Aortic Valve Replacement […]
CerraCap Ventures Invests in Acorai, Paving the Way for Transformative Advances in Heart Failure Management
COSTA MESA, Calif., Jan. 19, 2024 /PRNewswire/ — CerraCap Ventures, a leading venture capital firm in healthcare innovation, has invested in Acorai, a cutting-edge company in cardiovascular health technology. The investment will boost the development and deployment of Acorai’s novel…
ABBOTT ANNOUNCES FIRST GLOBAL PROCEDURES IN A CLINICAL TRIAL OF ITS VOLT™ PULSED FIELD ABLATION SYSTEM TO TREAT PATIENTS WITH ABNORMAL HEART RHYTHMS
ABBOTT PARK, Ill., Jan. 18, 2024 /PRNewswire/ — Abbott (NYSE: ABT) today announced the first global procedures have been conducted using the company’s new Volt™ Pulsed Field Ablation (PFA) System to treat patients battling common abnormal heart rhythms such as atrial fibrillation (AFib). Over 30 patients were recently treated in Australia as part of […]
Artivion Obtains $350 Million in Senior Secured Credit Facilities
Non-Dilutive Financing Provides Comprehensive Solution to Address Debt Maturities and Provide Flexibility to Further Optimize Capital Structure ATLANTA, Jan. 18, 2024 /PRNewswire/ — Artivion, Inc. (NYSE: AORT), a leading cardiac and vascular surgery company focused on aortic disease,…
HeartSciences Announces Results and Adjournment of Annual Shareholder Meeting
Southlake, TX, Jan. 18, 2024 (GLOBE NEWSWIRE) — Heart Test Laboratories, Inc. d/b/a HeartSciences (Nasdaq: HSCS; HSCSW) (“HeartSciences” or the “Company”), an artificial intelligence (AI)-powered medical technology company focused on transforming ECGs/EKGs to save lives through earlier detection of heart disease, announced today the results of the Company’s 2024 Annual […]
Nectero Medical Announces Initiation of Phase II/III Clinical Trial of the Nectero EAST® System for Treatment of Small- to Medium-Sized Abdominal Aortic Aneurysms
TEMPE, Ariz.–(BUSINESS WIRE)– #AbdominalAorticAneurysm–Nectero Medical, a clinical-stage biotechnology company pioneering novel therapies to treat aneurysmal disease and improve patients’ lives, today announced initiation of a Phase II/III clinical trial (stAAAble) to investigate the safety and efficacy of the Nectero Endovascular Aneurysm Stabilization Treatment (Nectero EAST®) System in patients with infrarenal abdominal aortic aneurysms (AAAs), maximum diameter 3.5 – 5.0cm. The Nectero EAST System is a single-use,
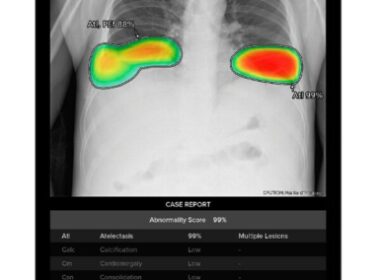
Lunit INSIGHT CXR Excels in Lung Nodule Detection – Exceptional Performance in Head-to-Head Study published in Radiology
– Lunit’s AI-powered chest X-ray analysis solution shows highest AUC of 0.93 in a recent multi-center, multi-use case, multi-vendor lung nodule detection study SEOUL, South Korea, Jan. 17, 2024 /PRNewswire/ — Lunit (KRX:328130.KQ), a leading provider of AI-powered solutions for cancer…
First U.S. Implantation of VasQ™ Since FDA De Novo was Granted
SPARTANBURG, S.C., Jan. 16, 2024 /PRNewswire/ — The first U.S. implantation of VasQ™ since FDA De Novo was granted was successfully performed by Dr. Ari Kramer, Director and principal surgeon of Vascular Access Surgery at Spartanburg Regional Hospital in Spartanburg, SC. Developed by…
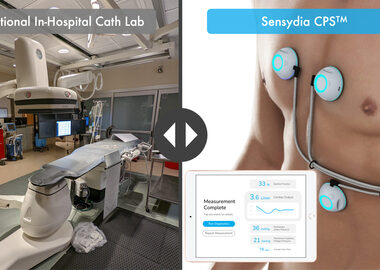
Sensydia Completes Fifth Study for Heart-Sound AI
LOS ANGELES–(BUSINESS WIRE)–Non-invasive cardiac assessment company Sensydia announced today that it has completed its 50-subject development study at the University of Minnesota (UMN). This study was conducted at UMN to collect data for its innovative AI-powered, non-invasive Cardiac Performance System (CPS™) that uses heart sound analysis to enable earlier detection and more effective therapy […]