KIRKLAND, Wash.–(BUSINESS WIRE)–Cardiac Dimensions®, a leader in the development of innovative and minimally invasive treatment modalities to address patients suffering from heart failure with functional mitral regurgitation (FMR), today announced that renowned interventional cardiologist Satya Shreenivas, MD, MBA has joined the company as Chief Medical Officer. “I have always been […]
Other News
IceCure Medical’s ProSense® Featured at Indian Society of Vascular and Interventional Radiology Conference: Key Opinion Leaders Highlight Benefits of Cryoablation
ProSense® users speaking at the conference about cryoablation included ISVIR President Dr. Suyash Kulkarni, as well as Dr. Shuvro Roy-Choudhury CAESAREA, Israel, Jan. 16, 2024 /PRNewswire/ — IceCure Medical Ltd. (Nasdaq: ICCM) (“IceCure” or the “Company”), developer of the ProSense®…
AM-Pharma Initiates Phase 2 Trial of Ilofotase Alfa for Cardiac Surgery-Associated Renal Damage
UTRECHT, The Netherlands, January 16, 2024–(BUSINESS WIRE)–AM-Pharma B.V. today announced that the first patients have been treated in a Phase 2 clinical study evaluating the company’s proprietary recombinant alkaline phosphatase, ilofotase alfa, as a preventive treatment for cardiac surgery-associated renal damage (CSA-RD). With over 2 million open heart surgeries1 performed worldwide every year, CSA-RD […]
Reflow Medical Receives CE Mark for Bare Temporary Spur Stent System for Treating de novo or Restenotic Below-the-Knee (BTK) Lesions
San Clemente, CA—Jan. 15, 2024 – Reflow Medical, Inc., a developer of innovative medical devices focused on cardiovascular disease, announces it has received CE (Conformité Européenne) Mark certification in the European Union for the Bare Temporary Spur Stent System. The device is intended to treat de novo or restenotic lesions […]
CARMAT announces a conditional agreement in principle with the EIB on new loan repayment terms, as part of the management of its short-term financing challenge
PARIS–(BUSINESS WIRE)–Regulatory News: “This agreement in principle with the EIB on new repayment terms for the loan is a positive signal for the Company. ” Post this CARMAT (FR0010907956, ALCAR, eligible for PEA-PME equity saving plans), designer and developer of the world’s most advanced total artificial heart, aiming to provide […]
InkSpace Imaging receives FDA Clearance for a 24-channel Small Body Array Compatible with Siemens Healthineers 3T Systems
PLEASANTON, Calif., Jan. 11, 2024 (GLOBE NEWSWIRE) — InkSpace Imaging, a leader in innovative diagnostic medical device technology, is proud to announce it received FDA clearance for its next-generation Small Body Array for the Siemens Healthineers Magnetom Skyra and Vida series 3T MRI scanners. This new medical device is part […]
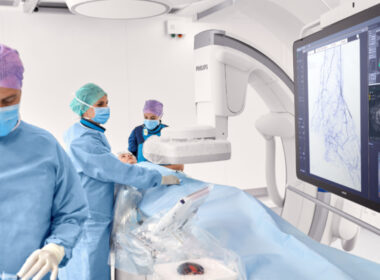
Multidisciplinary panel of experts from prominent cardiovascular societies advocate for broader adoption of IVUS in peripheral interventions to improve patient care
Amsterdam, the Netherlands – Royal Philips, a global leader in health technology, today announced that a group of multidisciplinary cardiovascular specialists from prominent medical societies have issued a joint expert opinion on the advantages of intravascular ultrasound (IVUS) use in peripheral arterial and deep venous interventions compared with using angiography alone. […]
Endovascular Engineering’s Hēlo™ Thrombectomy System Receives IDE Approval to Start ENGULF Pivotal Trial for the Treatment of Pulmonary Embolism
MENLO PARK, Calif., Jan. 11, 2024 /PRNewswire/ — Endovascular Engineering (“E2”), a medical device company focused on the development and deployment of groundbreaking clot removal technologies that target venous thromboembolism (VTE), announced today FDA Investigational Device Exemption…
United Imaging Intelligence Highlights Dedications to Advancing Intelligent Healthcare at RSNA Conference 2023
BOSTON, Jan. 11, 2024 /PRNewswire/ — The Radiological Society of North America (RSNA) has concluded its annual meeting in Chicago, IL, serving as a platform for revolutionary advancements in the field of radiology. United Imaging Intelligence (UII), an innovative subsidiary of United…
PulseAI’s Groundbreaking AI Algorithm Enhances Consumer ECG Diagnostics
Innovative AI Application Improves Cardiac Monitoring and Diagnostic Accuracy BELFAST, Northern Ireland , Jan. 11, 2024 /PRNewswire/ — PulseAI, a leader in artificial intelligence healthcare solutions, today announced a significant advancement in consumer device electrocardiogram (ECG)…