Global “DISSECT-N” Post-Market Study to Broaden Evidence Base for Safety and Effectiveness of Commercially Available Endovascular Repair Technology DUBLIN, July 06, 2020 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced the start of a prospective, observational, global, multi-center, real-world, post-market study to evaluate the safety and effectiveness of […]
Other News
MyoKardia Announces Appointment of Denelle J. Waynick as General Counsel and Corporate Secretary
BRISBANE, Calif., July 06, 2020 (GLOBE NEWSWIRE) — MyoKardia, Inc. (Nasdaq: MYOK) today announced the appointment of Denelle Waynick as General Counsel and Corporate Secretary. Ms. Waynick joins MyoKardia from UCB, Inc., bringing more than 25 years of domestic and international legal and business expertise in the healthcare and life-science […]
Endologix Enters into an Agreement with Deerfield Partners to Take the Company Private
Commences voluntary Chapter 11 process with access to $30.8 million in DIP financing Files consensual plan of reorganization that would reduce approximately $180 million in debt and provide an approximately $30 million in additional exit financing Confirms launch of Alto this summer, steady production, continued clinical studies, and accelerated pipeline […]
Carag Receives U.S. FDA IDE Approval to Conduct Clinical Study of First Transcatheter Septal Occluder With Bioresorbable, Metal-free Framework
The U.S. trial for CE-marked CBSO is designed to enroll up to 250 patients in a staged study approach BAAR, Switzerland, July 1, 2020 /PRNewswire/ — CARAG AG, a privately-held Swiss medical device development company, today announced receiving U.S. Food and Drug Administration (FDA) Investigational Device Exemption (IDE) approval for its Carag Bioresorbable […]
inHEART Raises $4.2 Million to Improve Treatments for Cardiac Arrhythmias With Medical Imaging, Artificial Intelligence and Numerical Simulations
PESSAC, France, July 2, 2020 /PRNewswire/ — inHEART, providing a cloud-based medical image analysis solution for cardiac interventions on patients with arrhythmias, has closed a round of EUR 3.7 million led by Elaia. These funds will be used to accelerate commercial development in Europe, access the US market, and advance its technology leadership with continued development of […]
Berlin Heart Completes Post Approval Surveillance; Report Details Improved Outcomes for Pediatric Heart Failure Patients Supported with EXCOR® Pediatric
THE WOODLANDS, Texas–(BUSINESS WIRE)–Berlin Heart Inc. announced they have completed the Post Approval Surveillance for the EXCOR® Pediatric Ventricular Assist Device, a requirement of the Post Market Approval (PMA) granted by the Food and Drug Administration (FDA), in June 2017. The final report submitted to the FDA confirmed the positive contemporary […]
Novartis Agrees to Pay $678 Million to Settle Allegations of Illegal Kickbacks Involving Several of the Company’s Cardiovascular Drugs
Attorney Eric L. Young Represents Whistleblower Oswald Bilotta in One of the Largest Ever Recoveries in a Health Care Fraud Case Brought Under the Qui Tam Provisions of the False Claims Act PHILADELPHIA, July 01, 2020 (GLOBE NEWSWIRE) — Attorney Eric. L. Young announced today that Novartis Pharmaceuticals Corporation (“Novartis”) has agreed to settle alleged […]
Dr. David G. Kallend joins DalCor Pharmaceuticals as Chief Scientific Officer
LONDON and MONTREAL, July 02, 2020 (GLOBE NEWSWIRE) — DalCor Pharmaceuticals today announced the appointment of Dr. David G. Kallend as Chief Scientific Officer effective July 1, 2020. Dr. Kallend joins DalCor from The Medicines Company where he held the position of Senior Vice President and Chief Medical Officer. He […]
Sofinnova Partners Announces Two First Investments from its MD Start III Medtech Acceleration Fund
Investments in innovative medical devices follow the fund’s initial closing in May 2019 PARIS–(BUSINESS WIRE)–Sofinnova Partners, a leading European life sciences venture capital firm based in Paris, London and Milan, announced today the second investment from its MD Start III Fund, a medtech accelerator that actively works with clinicians and […]
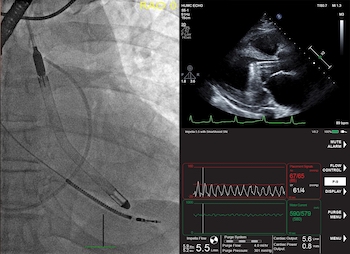
Study Finds 84% Survival Rate in Patients in Cardiogenic Shock and Other Challenging Cardiac Conditions with the New Impella 5.5 with SmartAssist
DANVERS, Mass.–(BUSINESS WIRE)–The first published United States experience of patients who received Abiomed’s newest heart pump, Impella 5.5 with SmartAssist, finds 84% of the patients survived to explant with 76% native heart recovery. The study was published in the July edition of the American Society for Artificial Internal Organs (ASAIO) Journal. The study examined […]