Global Study to Evaluate Percutaneous Device Designed for the Mechanical Support of the Ventricle in Heart Failure Patients SANTA CLARA, Calif.–(BUSINESS WIRE)–Ancora Heart, Inc., a company developing a novel therapy to address heart failure, today announced U.S. Food and Drug Administration (FDA) approval of its Investigational Device Exemption (IDE) application […]
Other News
CorFlow Reports Late-Breaking Data from the “MOCA I” Clinical Trial
BAAR, Switzerland–(BUSINESS WIRE)–CorFlow Therapeutics AG (“CorFlow”) announced today that Dr. Felice Gragnano, MD, Inselspital, Bern University Hospital, Bern, Switzerland, has presented interim clinical data from the MOCA I Phase 2 clinical trial during the PCR e-Course. The MOCA I study is a first-in-human (FIH) clinical trial to document the safety and feasibility of the CorFlow Controlled Flow […]
Verve Therapeutics Presents New Data in Non-Human Primates Validating Gene Editing as a Treatment Approach for Coronary Heart Disease at the ISSCR 2020 Virtual Annual Meeting
Data Demonstrate In Vivo Adenine Base Editing Can Turn Off Either PCSK9 or ANGPTL3 in the Liver and Substantially Lower LDL Cholesterol or Triglyceride Levels CAMBRIDGE, Mass.–(BUSINESS WIRE)–Verve Therapeutics, a next-generation cardiovascular company, today announced the presentation of new preclinical proof-of-concept data in non-human primates that demonstrate the successful use of base […]
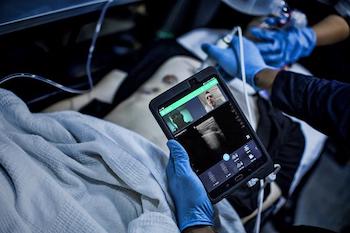
Philips expands its remote clinical collaboration offering based on the Reacts platform
Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced that it is expanding its remote clinical collaboration and virtual training offerings across its portfolio, building on the IIT * (Innovative Imaging Technologies) Reacts collaborative platform. Leveraging innovative technologies, such as augmented reality, for remote virtual guidance, […]
Intra-Cellular Therapies Announces Positive Top-line Results from ITI-214 Phase I/II Study in Patients with Heart Failure
Study demonstrated inhibiting phosphodiesterase-1 with ITI-214 in patients with heart failure improved cardiac function by enhancing cardiac contractility and dilating systemic arteries without inducing abnormal heart rhythms. NEW YORK, June 29, 2020 (GLOBE NEWSWIRE) — Intra-Cellular Therapies, Inc. (Nasdaq:ITCI) today announced topline results from Study ITI-214-104, a Phase I/II translational study […]
BioCardia Announces CardiAMP Heart Failure Pivotal Trial Continues with First Patient Randomized in COVID-19 Era
SAN CARLOS, Calif., June 29, 2020 (GLOBE NEWSWIRE) — BioCardia®, Inc., (OTC: BCDA), a leader in the development of comprehensive solutions for cardiovascular regenerative therapies, today announced that the Company has resumed cases in the CardiAMP Heart Failure Trial. The first patient procedure completed since elective procedures were paused due to […]
Neovasc Reducer™ and Tiara™ Featured at Leading Industry Virtual Conferences
VANCOUVER and MINNEAPOLIS, MN, June 29, 2020 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — Neovasc, Inc. (“Neovasc” or the “Company”) (NASDAQ, TSX: NVCN), today announced that the Neovasc Reducer™ (“Reducer”) and Tiara™ (“Tiara”) program were recently featured at two industry conferences. The Tiara Mitral Valve Replacement program was featured at both the PCR e-Course […]
Mezzion Announces Submission of New Drug Application for its Orphan Drug Udenafil to Treat Patients who have undergone the Fontan Operation for Single Ventricle Heart Disease
Submitted for FDA Priority Review as a Rare Pediatric Disease Product Application SEOUL, South Korea, June 30, 2020 /PRNewswire/ — Mezzion Pharma Co. Ltd. (140410.KQ), announced today that it has submitted a New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) for the approval of udenafil to improve the physiology of patients 12 […]
CVRx® Announces Publication of BeAT-HF Clinical Study Results in the Journal of The American College of Cardiology
BAROSTIM NEO™ is the World’s First Heart Failure Neuromodulation Device MINNEAPOLIS, June 30, 2020 /PRNewswire/ — CVRx, Inc., a private medical device company, announced today that its BeAT-HF phase III randomized clinical trial results were published in the Journal of the American College of Cardiology (“JACC”). Results from the trial were used […]
Manipal Hospitals Performs Quadruple Valve Procedure for the First Time in Karnataka Amidst Pandemic
BANGALORE, June 30, 2020 /PRNewswire/ — Manipal Hospitals Bangalore performed quadruple valve procedure for the first time in Karnataka to give new lease of life to 38-year-old male patient. Dr. Devananda N S, HOD & Consultant – Cardiothoracic Vascular Surgery, Heart and Lung Transplant Surgery, Manipal Hospitals Old Airport Road, performed this […]