Atrioventricular Interval Modulation (“AVIM”) Therapy Food and Drug Administration (“FDA”) Breakthrough Device Designations span the broader population of patients with uncontrolled hypertension despite medication at increased cardiovascular risk and the specific pacemaker-indicated population being evaluated in the BACKBEAT TrialThe addressable U.S. patient population for AVIM Therapy, based on the indications for use specified in its Breakthrough Device Designations, comprises over 7.7 million adults with hypertension despite medicationBreakthrough Device Designation supports favorable reimbursement pathways, including potential eligibility for New Technology Add-on Payment (“NTAP”) and Transitional Pass-Through (“TPT”) payment, which can facilitate broader, more timely patient access and provider adoption NEW HOPE, Pa., April 30, 2026 (GLOBE NEWSWIRE) — Orchestra BioMed Holdings, Inc. (Nasdaq: OBIO) (“Orchestra BioMed” or the “Company”), a biomedical company accelerating high-impact technologies to patients through strategic partnerships with market-leading global medical device companies, today announced that the FDA has granted a second Breakthrough Device Designation (“BDD”) for AVIM Therapy specific to patients with uncontrolled hypertension despite the use of anti-hypertensive medications, and an indication for a pacemaker. Together, the two BDDs for AVIM Therapy cover indications that encompass both the broader population of patients with uncontrolled hypertension despite medication and increased cardiovascular risk as well as the specific pacemaker-indicated population with uncontrolled hypertension being evaluated in the BACKBEAT Global Pivotal Trial (“BACKBEAT Trial”), which Orchestra BioMed is conducting in collaboration with Medtronic (NYSE: MDT). This additional BDD supports strategic optionality for the clinical, regulatory and commercial reimbursement strategies for AVIM Therapy for both the pacemaker population and potential future expansion populations with uncontrolled hypertension and increased cardiovascular risk. “We are pleased to receive this additional Breakthrough Device Designation from the FDA. We believe it is directly aligned with the patient population being studied in our ongoing BACKBEAT Trial which we are executing in collaboration with Medtronic,” said David Hochman, Chairman and Chief Executive Officer of Orchestra BioMed. “Continued alignment from the FDA, alongside our strategic relationship with Medtronic and accelerated enrollment in the BACKBEAT Trial, puts us in a strong position to advance AVIM Therapy to pivotal trial results.” Orchestra BioMed has a strategic collaboration with Medtronic, the global market leader in cardiac pacing therapies, for development and commercialization of AVIM Therapy for the treatment of uncontrolled hypertension in pacemaker-indicated patients. Under the terms of the existing collaboration agreement, Medtronic holds the right of first negotiation to expand its licensing agreement with Orchestra BioMed to obtain global rights to commercialize AVIM Therapy for the treatment of uncontrolled hypertension in patients that do not have an indication for a pacemaker. The FDA Breakthrough Devices Program, which reflects the FDA’s commitment to device innovation and protecting public health, is designed to expedite the development of and provide priority review for innovative medical technologies that have the potential to significantly improve outcomes for patients with serious or life-threatening conditions. To be eligible for this designation, a device must demonstrate the potential to provide more effective treatment or diagnosis of a life-threatening or irreversibly debilitating condition. In addition, the device must meet at least one of the following criteria: it must represent breakthrough technology, have no approved or cleared alternatives, offer significant advantages over existing options, or be determined by the FDA to be in the best interest of patients. Beyond regulatory acceleration, the BDD may also support favorable reimbursement pathways, including eligibility for incremental inpatient reimbursement through the New Technology Add-on Payment and outpatient Transitional Pass-Through payments under the Centers for Medicare & Medicaid Services programs. These mechanisms may help facilitate more timely access to breakthrough technologies while supporting provider adoption and patient access. About Orchestra BioMed Orchestra BioMed is a biomedical innovation company accelerating high-impact technologies to patients through strategic collaborations with market-leading global medical device companies. The Company’s two flagship product candidates – Atrioventricular Interval Modulation (AVIM) Therapy and Virtue® Sirolimus AngioInfusion™ Balloon (Virtue SAB) – are currently undergoing pivotal clinical trials for their lead indications, each representing multi-billion-dollar annual global market opportunities. AVIM Therapy is a bioelectronic treatment for hypertension, the leading risk factor for death worldwide, and is designed to be delivered by a pacemaker and achieve immediate, substantial and sustained reductions in blood pressure in patients with hypertensive heart disease. The Company has a strategic collaboration with Medtronic, one of the largest medical device companies in the world and the global leader in cardiac pacing therapies, for the development and commercialization of AVIM Therapy for the treatment of uncontrolled hypertension in pacemaker-indicated patients. AVIM Therapy has FDA Breakthrough Device Designations for these patients, as well as an estimated 7.7 million total patients in the U.S. with uncontrolled hypertension despite medical therapy and increased cardiovascular risk. Virtue SAB is a highly differentiated, first-of-its-kind non-coated drug delivery angioplasty balloon system designed to deliver a large liquid dose of proprietary extended-release formulation of sirolimus, SirolimusEFR™, for the treatment of atherosclerotic artery disease, the leading cause of mortality worldwide. Virtue SAB has been granted Breakthrough Device Designation by the FDA for the treatment of coronary in-stent restenosis, coronary small vessel disease and below-the-knee peripheral artery disease. For further information about Orchestra BioMed, please visit www.orchestrabiomed.com, and follow us on LinkedIn. About AVIM TherapyAVIM Therapy is an investigational therapy compatible with standard dual-chamber pacemakers designed to substantially and persistently lower blood pressure. It has been evaluated in pilot studies in patients with hypertension who are also indicated for a pacemaker. MODERATO II, a double-blind, randomized pilot study, showed that patients treated with AVIM Therapy experienced net reductions of 8.1 mmHg in 24-hour ambulatory systolic blood pressure (aSBP) and 12.3 mmHg in office systolic blood pressure (oSBP) at six months when compared to control patients. In addition to reducing blood pressure, clinical results using AVIM Therapy demonstrate improvements in cardiac function and hemodynamics. The BACKBEAT (BradycArdia paCemaKer with atrioventricular interval modulation for Blood prEssure treAtmenT) global pivotal study will evaluate the safety and efficacy of AVIM Therapy in lowering blood pressure in patients who have systolic blood pressure above target despite anti-hypertensive medication and who are indicated for or have recently received a dual-chamber cardiac pacemaker. AVIM Therapy has been granted two Breakthrough Device Designations by the FDA for the treatment of uncontrolled hypertension in patients who have increased cardiovascular risk. Forward-Looking Statements Certain statements included in this press release that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995. Forward-looking statements generally are accompanied by words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “seem,” “seek,” “future,” “outlook” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements relating to the initiation, enrollment, timing, implementation and design of the Company’s ongoing pivotal trials, realizing the clinical and commercial value of AVIM Therapy and Virtue SAB, the potential safety and efficacy of the Company’s product candidates, the potential benefits of BDD, including its ability to expedite FDA reviews and support favorable reimbursement pathways, and the ability of the Company’s partnerships to accelerate clinical development. These statements are based on various assumptions, whether or not identified in this press release, and on the current expectations of the Company’s management and are not predictions of actual performance. These forward-looking statements are provided for illustrative purposes only and are not intended to serve as and must not be relied on as a guarantee, an assurance, a prediction, or a definitive statement of fact or probability. Actual events and circumstances are difficult or impossible to predict and may differ from assumptions. Many actual events and circumstances are beyond the control of the Company. These forward-looking statements are subject to a number of risks and uncertainties, including changes in domestic and foreign business, market, financial, political, and legal conditions; risks related to regulatory approval of the Company’s commercial product candidates and ongoing regulation of the Company’s product candidates, if approved; the timing of, and the Company’s ability to achieve expected regulatory and business milestones; the impact of competitive products and product candidates; and the risk factors discussed under the heading “Item 1A. Risk Factors” in the Company’s Annual Report on Form 10-K for the year ended December 31, 2025, which was filed with the SEC on March 12, 2026. The Company operates in a very competitive and rapidly changing environment. New risks emerge from time to time. Given these risks and uncertainties, the Company cautions against placing undue reliance on these forward-looking statements, which only speak as of the date of this press release. The Company does not plan and undertakes no obligation to update any of the forward-looking statements made herein, except as required by law. Investor Contact:Silas NewcombOrchestra BioMedSnewcomb@orchestrabiomed.com Media Contact:Kelsey KirkOrchestra BioMedkkirkellis@orchestrabiomed.com
Coronary/Structural Heart
Boehringer Ingelheim and Eko Health Inc. launch new AI-based solution that detects heart murmurs in dogs
Eko Vet+TM | CANINEBEAT® AI helps veterinarians detect, visualize, and grade heart murmurs in dogs that can otherwise go undetectedThe CANINEBEAT® AI algorithm detects heart murmurs associated with structural heart disease in dogs with more than 95% sensitivity and specificity*With earlier murmur detection, dogs can benefit from heart disease diagnostics and treatment sooner INGELHEIM, Germany, and SAN FRANCISCO, US – Boehringer Ingelheim, a global leader in animal health, and Eko Health, a leader in AI-powered cardiac and pulmonary disease detection, announced the launch of an innovative solution to detect, visualize, and grade heart murmurs in dogs. Eko Vet+TM | CANINEBEAT® AI aims to improve health outcomes for dogs with heart disease by enabling veterinarians to identify heart murmurs that might otherwise go unnoticed. “Our proprietary CANINEBEAT® AI algorithm integrated into the Eko Vet+TM app will powerfully support veterinarians’ early detection of heart murmurs in dogs and pet owner conversations,” said Dr. Erich Schött, Head of the Pet Business at Boehringer Ingelheim. “Heart murmur detection in dogs can be quite difficult, especially in busy clinics. Because pet owners don’t see clinical signs in the early stage of heart disease, it’s a challenge for veterinarians to explain that the dog suffers from a serious disease. With this unique AI solution, we can help detect murmurs earlier in the disease process, so dogs may receive appropriate diagnosis and treatment sooner.” The new digital solution includes three integrated components: ● The Eko CORE™ Digital Attachment connects to most single-tube stethoscopes and digitizes heart sounds that can then be analyzed by AI-based algorithms. It also helps veterinarians hear more subtle murmurs by amplifying heart sounds by 40 times and offers advanced noise cancellation and cardiac sound filtering. Other Eko devices also are compatible with the veterinary digital solution. ● The CANINEBEAT® AI algorithm, trained and validated on more than 4,000 annotated canine heart sound recordings, detects heart murmurs associated with structural heart disease in dogs with more than 95% sensitivity and specificity.* The algorithm is intended to support veterinary clinical assessment and does not replace comprehensive cardiac evaluation or professional veterinary judgment.● The EkoVet+TM app incorporates insights from the CANINEBEAT® AI algorithm, including murmur images, sound files, and shareable reports. The app provides clinical support and helps veterinarians discuss the potential findings and any diagnostics and treatment needs with pet owners. Heart disease affects approximately 10% of all dogs.1 The most common heart disease in dogs is myxomatous mitral valve disease (MMVD), which often produces a heart murmur when the heart has a leak in one of the heart valves, preventing blood from getting pumped correctly. MMVD is a chronic condition that is a major cause of morbidity and mortality in dogs.2 Because dogs in early stages of heart disease may be asymptomatic, heart murmurs can go unnoticed during routine examinations. To address this challenge, 50 global veterinary cardiology experts contributed to the development of Eko Vet+TM with CANINEBEAT® AI. “CANINEBEAT® AI inside Eko Vet+TM is an exceptional resource for general practitioner veterinarians, helping them quickly detect and grade murmurs with a high level of consistency,” said Prof. Gerhard Wess, Head of the Cardiology Service, Clinic for Small Animal Internal Medicine, Ludwig-Maximilians-Universität in Munich. “I am thrilled to be involved in the development of this new AI-powered solution that will give veterinarians greater confidence in heart murmur detection and support better outcomes for dogs.” The introduction of this innovative auscultation solution for the veterinary industry has begun in the United States and the United Kingdom and will start in Germany next month. A phased expansion is planned in additional markets later this year and next. It will be available through both Boehringer Ingelheim and Eko Health, comprising the same three components through distinct commercial offers. “We’ve seen firsthand in human health how earlier, more accurate detection of cardiac disease can change the trajectory of care,” said Connor Landgraf, CEO and co-founder of Eko Health. “Collaborating with Boehringer Ingelheim, a leader in canine cardiology, allows us to bring that same innovative approach to veterinary medicine. Together, we’re equipping veterinarians with AI-powered tools to identify heart murmurs earlier and support better outcomes in canine patients.” About Boehringer Ingelheim – Animal Health business Boehringer Ingelheim provides innovation for preventing and treating diseases in animals. The company offers a wide range of vaccines, parasite-control products, and medicines for pets, horses, and livestock to veterinarians, animal owners, farmers, and governments. As a leader in animal health, Boehringer Ingelheim values that the health of humans and animals is deeply connected and strives to make a difference for people, animals, and society. Learn more at www.boehringer-ingelheim.com/animal-health. About Boehringer IngelheimBoehringer Ingelheim is a biopharmaceutical company active in both human and animal health. As one of the industry’s top investors in research and development, the company focuses on developing innovative therapies that can improve and extend lives in areas of high unmet medical need. Independent since its foundation in 1885, Boehringer takes a long-term perspective, embedding sustainability along the entire value chain. Our approximately 54,300 employees serve over 130 markets to build a healthier and more sustainable tomorrow. Learn more at www.boehringer-ingelheim.com. About Eko HealthEko Health is a pioneering AI early disease detection company advancing how healthcare professionals detect and monitor heart and lung disease with its portfolio of digital stethoscopes, ECG devices, software, and AI-powered analysis. Its FDA-cleared platform, with more than 700,000 devices sold worldwide, helps clinicians detect earlier and manage treatment more effectively to improve patient outcomes. Eko Health is headquartered in Emeryville, California. For more information, visit www.ekohealth.com. Media Contacts:Mi-Kyung Lee Langemi-kyung.lee-lange@boehringer-ingelheim.com Sam Moore sam.moore@ekohealth.com Editorial Note: * The CANINEBEAT study was conducted using one of the largest canine digital auscultation datasets to date, comprising more than 4,000 heart sound recordings collected from over 3,400 dogs. The study assessed the performance of the CANINEBEAT® AI algorithm in detecting canine heart murmurs. All recordings were systematically annotated, with the algorithm’s development and validation supported by nearly 50 veterinary cardiology experts. Detailed study results will be presented and published in peer reviewed scientific forums. References: 1 Keene, Bruce W et al. “ACVIM consensus guidelines for the diagnosis and treatment of myxomatous mitral valve disease in dogs.” Journal of veterinary internal medicine vol. 33,3 (2019): 1127-1140. https://doi.org/10.1111/jvim.154882 Häggström, J et al. “New insights into degenerative mitral valve disease in dogs.” Veterinary Clinics of North America: Small Animal Practice. 2004 vol 34, 5: 1209-1226. doi: 10.1016/J.CVSM.2004.05.002.
CorVista Health Announces Collaboration with Mayo Clinic to Advance Next-Gen Pulmonary Hypertension Diagnosis
BETHESDA, Md.–(BUSINESS WIRE)–CorVista Health has announced a research collaboration with Mayo Clinic to evaluate how next-generation, non-invasive diagnostics may transform the identification and management of pulmonary hypertension (PH) – a complex and frequently underdiagnosed cardiovascular condition that affects an estimated one percent of the global population. Pulmonary hypertension is a progressive and potentially life-threatening condition characterized by elevated pressure in the pu
BridgeBio Launches Health Education Effort on Often Overlooked Heart Condition Featuring Attruby® Advocates, Morgan Freeman and Sports Business Icon Howard H. White
– New national campaign encourages recognition of ATTR-CM, often mistaken for other heart conditions, where earlier diagnosis and treatment may help improve outcomes1 PALO ALTO, Calif., April 28, 2026 (GLOBE NEWSWIRE) — BridgeBio Pharma, Inc. (Nasdaq: BBIO) (the “Company,” “we” or “BridgeBio”), a biopharmaceutical company focused on developing medicines for genetic conditions, has teamed up with award-winning actor Morgan Freeman and Howard “H” White, a longtime basketball apparel marketing executive, cultural connector, and heart health advocate, to bring greater attention to ATTR-CM, a progressive heart disease that can be underrecognized and often mistaken for other forms of heart failure.1,2 Don’t Pass On Your Heart Health encourages people to take persistent symptoms seriously, recognizing that earlier action can help support better long-term outcomes. Although ATTR-CM has historically been diagnosed more often in men, research suggests the need for broader awareness across men and women over 55.3 “In clinical practice, ATTR-CM is often hiding in plain sight. Many people come in with symptoms like shortness of breath, fatigue, carpal tunnel, nerve pain, or leg swelling that are commonly attributed to other forms of heart failure or aging,” said Albert J. Hicks, III, M.D., MPH, Section Chief of Heart Failure & Cardiac Transplant at University of Maryland Medical Center. “What’s critical is recognizing when those symptoms may point to an underlying condition that requires further evaluation. Increasing awareness among both clinicians and patients can help prompt earlier recognition, which is an important step toward improving long-term outcomes.” “We continue to see a disproportionate impact of ATTR-CM in Black communities, where people may face delays in diagnosis and access to care,” said Richard Allen Williams, M.D., FACC, Founder and past President of the Association of Black Cardiologists. “Improving awareness is critical, but so is ensuring people feel empowered to speak up, ask questions, and advocate for themselves when something doesn’t feel right.” Howard “H” White is known for shaping athlete-driven movements in global sports culture. Over a four-decade marketing career, he is an author, philanthropist and is widely recognized for building connections between sports, community, and culture, including founding youth-focused initiatives that empower the next generation. Howard experienced symptoms not typically associated with heart failure or heart disease, including carpal tunnel syndrome and an enlarged heart. As his condition progressed, he developed shortness of breath, leg swelling, and fatigue, and by the time he recognized the seriousness of his symptoms, his disease had significantly advanced. He is now committed to ensuring others do not go through the same experience, encouraging anyone experiencing these symptoms to speak with their doctor early so it does not progress to that point. His commitment to advancing awareness and care also includes founding the Howard “H” White Center for Cardiac Amyloidosis at Providence St. Vincent Medical Center, which focuses on improving diagnosis, treatment, and education around amyloid heart disease. “For years, people have been living with cardiac amyloidosis without knowing it,” said Howard “H” White. “I didn’t recognize it until simple things – like walking up the stairs – left me out of breath. I had been told my enlarged heart was from sports. The truth was, amyloid was making my heart bigger and weaker. By the time I found out, my condition had already progressed significantly. Today, earlier detection is possible. My message is simple: know the signs, ask questions, and see your doctor so you don’t have to follow my path.” Transthyretin (TTR) is a protein that circulates in the blood, where it transports thyroxine and vitamin A and supports critical functions, including cognition, cardiovascular health, muscle integrity, metabolism, vision, and bone strength. ATTR-CM occurs when misfolded TTR protein, also known as amyloid, builds up in the heart over time. As deposits accumulate, the heart can become thicker and stiffer, making it harder to pump blood effectively. Because symptoms may resemble those of more common forms of heart failure, or be dismissed as “normal aging,” ATTR-CM is frequently overlooked or identified later in the disease journey.3 ATTR-CM includes both wild-type disease associated with aging and hereditary (variant) forms caused by genetic changes in the transthyretin (TTR) gene.1 Certain variants are more commonly carried by people of African ancestry, and approximately 1 in 25 Black/African American individuals in the United States carry the V142I (also known as V122I) variant associated with ATTR-CM.2 As a result, Black men and women over 55 may face elevated risk and delayed diagnosis, with research showing that people with this variant can experience more severe disease and poorer outcomes, including worse survival and higher rates of heart failure.2,4,5 Recent clinical findings highlight why recognizing ATTR-CM earlier can make a meaningful difference. In the Phase 3 ATTRibute-CM study evaluating Attruby®, the treatment showed consistent benefit in people with both wild-type and hereditary forms of the disease.6,7 Changes linked to stabilizing the TTR protein were seen as early as four weeks after starting treatment and were sustained over time.8 Over the course of the study, Attruby helped adults with ATTR-CM live longer and have fewer hospitalizations due to heart issues. 9 At 30 months, treatment was associated with a 50% reduction in the cumulative frequency of cardiovascular-related hospitalizations compared to placebo, and people taking Attruby also reported a better health-related quality of life compared to those not taking Attruby.6,7,10 The majority of side effects, like diarrhea and abdominal pain, were mild and went away on its own, allowing people to stay on Attruby.9 “ATTR-CM is a condition where earlier awareness can make a meaningful difference, yet many people are still diagnosed only after symptoms have progressed,” said Jonathan Fox, M.D., Ph.D., Chief Medical Officer at BridgeBio Cardiovascular. “This is particularly important for individuals carrying genetic variants, such as p.Val142Ile (also known as V122I), who often experience delays in diagnosis and limited awareness, which can contribute to poorer outcomes over time. Improving recognition of the disease, especially at a stage when intervention may help stabilize disease progression, is an important step toward better long-term outcomes.” To support people in taking the next step for their heart health, BridgeBio has launched a new educational hub on Attruby.com offering clear information about ATTR-CM including Howard “H” White’s testimonial video, practical resources, and a downloadable Doctor Discussion Guide to help individuals and families recognize potential warning signs and prepare for informed conversations with healthcare providers. BridgeBio continues research to better understand outcomes in both wild-type and variant ATTR-CM, helping to advance knowledge of how the disease affects different populations. To hear more from Howard “H” White on the importance of recognizing ATTR-CM earlier, visit: attruby.com/howard-h-white About Attruby® (acoramidis) INDICATIONAttruby is a transthyretin stabilizer indicated for the treatment of the cardiomyopathy of wild-type or variant transthyretin-mediated amyloidosis (ATTR-CM) in adults to reduce cardiovascular death and cardiovascular-related hospitalization.7 IMPORTANT SAFETY INFORMATION Adverse ReactionsDiarrhea (11.6% vs 7.6%) and upper abdominal pain (5.5% vs 1.4%) were reported in patients treated with Attruby versus placebo, respectively. The majority of these adverse reactions were mild and resolved without drug discontinuation. Discontinuation rates due to adverse events were similar between patients treated with Attruby versus placebo (9.3% and 8.5%, respectively).7 About BridgeBio Pharma, Inc.BridgeBio exists to develop transformative medicines for genetic conditions. Millions of people worldwide living with genetic conditions lack treatment options, often because drug development for small patient populations can be commercially challenging. We aim to bridge the gap between advancements in genetic science and meaningful medicines for underserved patient populations. Our decentralized, hub-and-spoke model is designed for speed, precision, and scalability. Autonomous and empowered teams focus on individual conditions, while a central hub provides the clinical, regulatory, and commercial capabilities needed to bring innovation to market. For more information, visit bridgebio.com and follow us on LinkedIn, X, Facebook, Instagram, YouTube, and TikTok. BridgeBio Pharma, Inc. Forward-Looking StatementsThis press release contains forward-looking statements. Statements in this press release may include statements that are not historical facts and are considered forward-looking within the meaning of Section 27A of the Securities Act of 1933, as amended (the Securities Act), and Section 21E of the Securities Exchange Act of 1934, as amended (the Exchange Act), which are usually identified by the use of words such as “anticipates,” “believes,” “continues,” “estimates,” “expects,” “hopes,” “intends,” “may,” “plans,” “projects,” “remains,” “seeks,” “should,” “will,” and variations of such words or similar expressions. BridgeBio intends these forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act and Section 21E of the Exchange Act. These forward-looking statements include statements regarding the potential benefits of earlier awareness, recognition, diagnosis and treatment of ATTR-CM, the potential ability of intervention to help stabilize disease progression or improve long-term outcomes, and the Company’s ongoing and future research regarding outcomes in wild-type and variant ATTR-CM. Although the Company believes that its plans, intentions, expectations and strategies as reflected in or suggested by those forward-looking statements are reasonable, the Company can give no assurance that the plans, intentions, expectations or strategies will be attained or achieved. Furthermore, actual results may differ materially from those described in the forward-looking statements and will be affected by a number of risks, uncertainties and assumptions, including, but not limited to those risks set forth in the Risk Factors section of the Company’s most recent Annual Report on Form 10-K and the Company’s other filings with the U.S. Securities and Exchange Commission. Moreover, the Company operates in a very competitive and rapidly changing environment in which new risks emerge from time to time. These forward-looking statements are based upon the current expectations and beliefs of the Company’s management as of the date of this press release, and are subject to certain risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Except as required by applicable law, BridgeBio assumes no obligation to update publicly any forward-looking statements, whether as a result of new information, future events or otherwise. BridgeBio Contact:Bubba Murarka, Executive Vice Presidentcontact@bridgebio.com (650)789-8220 References: Attruby.com, “What Is ATTR-CM?” Disease overview and symptom information. Accessed January 27, 2026.Attruby.com, “Signs & Symptoms” section. Accessed January 27, 2026.Buxbaum JN, Ruberg FL, Tripp KP, et al. The amyloidogenic V122I transthyretin variant in elderly Black Americans. N Engl J Med. 2014;370(23):2135–2142. Accessed January 27, 2026.Khedraki R, et al. Race, genotype, and prognosis in transthyretin amyloid cardiomyopathy. Am J Cardiol. 2024;216:97–104. doi:10.1016/j.amjcard.2024.02.019. Accessed March 24, 2026.Damrauer SM, Chaudhary K, Cho JH, et al. Association of the TTR V122I Variant With Heart Failure Among Individuals of African or Hispanic/Latino Ancestry. JAMA. 2019;322(22):2191–2202. doi:10.1001/jama.2019.17935. Accessed March 24, 2026Gillmore JD, Judge DP, Cappelli F, et al. Efficacy and safety of acoramidis in transthyretin amyloid cardiomyopathy. N Engl J Med. 2024;390(2):132-142.doi:10.1056/NEJMoa2305434.Attruby® (acoramidis) Prescribing Information. BridgeBio Pharma, Inc. Accessed January 27, 2026.Maurer M, Sarswat N, Grogan M, et al. Acoramidis improves serum TTR levels in patients with wild-type or variant transthyretin amyloid cardiomyopathy–results from ATTRibute-CM. Poster presented at: Annual Congress of the Heart Failure Association of the European Society of Cardiology; May 17-20, 2025; Belgrade, Serbia. Accessed March 24, 2026.BridgeBio Pharma, Inc. (2024). Acoramidis Significantly Reduces All-cause Mortality in the Overall ATTR-CM Variant and V142I (V122I) Populations press release. Accessed February 3, 2026.BridgeBio Pharma, Inc. (2025). Acoramidis Demonstrates Statistically Significant Reduction in Cardiovascular Mortality (CVM) Through Month 42 of the ATTRibute-CM Open-Label Extension. August 30, 2025. Available at: investor.bridgebio.com. Accessed March 30, 2026.
Anteris Technologies Secures CMS Reimbursement Supporting U.S. Site Activation for PARADIGM Trial
MINNEAPOLIS and BRISBANE, Australia, April 28, 2026 (GLOBE NEWSWIRE) — Anteris Technologies Global Corp. (“Anteris” or the “Company”) (NASDAQ: AVR, ASX: AVR), a global structural heart company committed to designing, developing, and commercializing cutting-edge medical devices to restore healthy heart function, today announced it has secured U.S. Medicare reimbursement eligibility for the global pivotal PARADIGM Trial under a Centers for Medicare & Medicaid Services (CMS) national coverage policy. Eligible procedures performed at participating U.S. study sites are covered under the Transcatheter Aortic Valve Replacement (TAVR) National Coverage Determination 20.32. The CMS framework operates under a Coverage with Evidence Development (CED) model, enabling reimbursement with clinical evidence generation in the PARADIGM Trial. This milestone is expected to facilitate U.S. site activation, accelerating operational momentum across participating centers. About the PARADIGM Trial The PARADIGM Trial is a prospective randomized controlled trial which will evaluate the safety and effectiveness of the DurAVR® Transcatheter Heart Valve (“THV”) compared to commercially available transcatheter aortic valve replacements (TAVRs). This head-to-head study will enroll approximately 1000 patients in the ‘All Comers Randomized Cohort’ with 1:1 randomization of patients who will receive either the DurAVR® THV or TAVR using commercially available and approved THVs. The PARADIGM Trial will assess non-inferiority on a primary composite endpoint of all-cause mortality, all stroke and cardiovascular hospitalization at one year post procedure. For further information, please refer to ClinicalTrials.gov NCT07194265. About Anteris Anteris Technologies Global Corp. (NASDAQ: AVR, ASX: AVR) is a global structural heart company committed to designing, developing, and commercializing cutting-edge medical devices to restore healthy heart function. Founded in Australia, with a significant presence in Minneapolis, USA, Anteris is a science-driven company with an experienced team of multidisciplinary professionals delivering restorative solutions to structural heart disease patients. Anteris’ lead product, the DurAVR® THV, was designed in collaboration with the world’s leading interventional cardiologists and cardiac surgeons to treat aortic stenosis – a potentially life-threatening condition resulting from the narrowing of the aortic valve. The balloon-expandable DurAVR® THV is the first biomimetic valve, which is shaped to mimic the performance of a healthy human aortic valve and aims to replicate normal aortic blood flow. DurAVR® THV is made using a single piece of molded ADAPT® tissue, Anteris’ patented anti-calcification tissue technology. ADAPT® tissue, which is FDA-cleared, has been used clinically for over 10 years and distributed for use in over 55,000 patients worldwide. The DurAVR® THV System is comprised of the DurAVR® valve, the ADAPT® tissue, and the balloon-expandable ComASUR® Delivery System. Forward-Looking Statements This announcement contains forward-looking statements, including statements regarding the expectation that achievement of the milestone will facilitate U.S. site activation, accelerating operational momentum across participating centers, and the PARADIGM Trial. Forward-looking statements include all statements that are not historical facts. Forward-looking statements generally are identified by the words “believe,” “project,” “expect,” “anticipate,” “estimate,” “intend,” “budget,” “target,” “aim,” “strategy,” “plan,” “guidance,” “outlook,” “may,” “should,” “could,” “will,” “would,” “will be,” “will continue,” “will likely result” and similar expressions, although not all forward-looking statements contain these identifying words. These forward-looking statements are subject to a number of risks, uncertainties, and assumptions, including those described under “Risk Factors” in Anteris’ Annual Report on Form 10-K for the fiscal period ended December 31, 2025 that was filed with the Securities and Exchange Commission and ASX. Readers are cautioned not to put undue reliance on forward-looking statements, and except as required by law, Anteris does not assume any obligation to update any of these forward-looking statements to conform these statements to actual results or revised expectations. For more information: Global Investor Relations Investor Relations (US)investors@anteristech.commchatterjee@bplifescience.comDebbie Ormsby Malini Chatterjee, Ph.D. Anteris Technologies Global Corp.Blueprint Life Science Group+61 1300 550 310 | +61 7 3152 3200+1 917 330 4269 Website www.anteristech.comX@AnterisTechLinkedInhttps://www.linkedin.com/company/anteristech
Tenaya Therapeutics Announces Late-Breaking Oral Presentation of New Clinical Data from RIDGE™-1 Phase 1b/2 Clinical Trial of TN-401 Gene Therapy in Adults with PKP2-Associated ARVC at ASGCT 2026
Readout to Include One-Year Cohort 1 Results and Early Cohort 2 Data; Webcast Conference Call Planned to Review RIDGE-1 Data at ASGCT Additional Presentations at ASGCT Showcase Tenaya’s Work with Patients to Advance Gene Therapy and Early-Stage Pipeline Innovations in Cardiac Gene Editing SOUTH SAN FRANCISCO, Calif., April 27, 2026 (GLOBE NEWSWIRE) — Tenaya Therapeutics, Inc. (NASDAQ: TNYA), a clinical-stage biotechnology company with a mission to discover, develop and deliver potentially curative therapies that address the underlying causes of heart disease, today announced the acceptance of multiple abstracts for presentation at the American Society of Gene and Cell Therapy (ASGCT) Annual Meeting, taking place May 11-15, 2026, in Boston, Massachusetts. Of note, new clinical data from both dose cohorts of the RIDGE-1 Phase 1b/2 trial of TN-401 will be featured as a late-breaking oral presentation. TN-401 is being developed for the potential treatment of adults with arrhythmogenic right ventricular cardiomyopathy (ARVC), a form of arrhythmogenic cardiomyopathy (ACM) that primarily impacts the right ventricle, caused by mutations in the plakophilin-2 (PKP2) gene. PKP2 gene mutations result in insufficient levels of critical proteins needed to maintain the structural integrity and cell-to-cell electrical signaling of heart muscle cells. TN-401 gene replacement therapy is designed to address the underlying cause of disease by delivering a functional PKP2 gene into heart muscle cells using an adeno associated virus serotype 9 (AAV9) capsid. The ASGCT presentation will include new safety, biopsy and efficacy data from patients treated at both the 3E13 vg/kg and 6E13 vg/kg dose levels. Details of the TN-401 clinical data presentation are as follows: Presentation Date & Time: Friday, May 15, 2026, from 8:00 am – 9:00 am EDT Abstract Title: Interim Data from RIDGE-1: A Phase 1b/2 Interventional Study to Evaluate Safety and Efficacy of TN-401, an AAV9 Investigational Gene Replacement Therapy, in Adults with PKP2-Associated Arrhythmogenic Right Ventricular Cardiomyopathy Location: Westin Seaport Commonwealth Ballroom ABC (Concourse Level)Presenting Author: John Giudicessi, M.D., Departments of Cardiovascular Medicine and Molecular Pharmacology and Experimental Therapeutics, Mayo Clinic, Rochester, New York Two other Tenaya abstracts have been accepted for poster presentations during ASGCT 2026. The first poster will detail results of a survey exploring parental perceptions of gene therapy treatment for children with cardiomyopathies conducted in partnership with DDC Clinic and the patient advocacy group, Children’s Cardiomyopathy Foundation. Presentation Session Date & Time: Tuesday, May 12, 2026, from 5:00 pm – 6:30 pm EDT Abstract Title: Perceptions and attitudes towards gene replacement therapy in parents of children with cardiomyopathy (#1372)Location: MCEC Exhibit and Poster HallPresenting Author: Kimberly Cohee, Executive Director, Patient Advocacy at Tenaya The second poster accepted for presentation builds on research previously presented for TN-501, a gene editing therapeutic candidate intended for the treatment of PLN-R14del-associated dilated cardiomyopathy (DCM). TN-501 is uniquely designed to specifically inactivate the pathogenic phosolamban (PLN) R14del allele while preserving healthy function. Presentation Session Date & Time: Thursday, May 14, 2026, from 5:00 pm – 6:30 pm EDTAbstract Title: Development of TN-501, an AAV-Delivered Gene Editing Therapy for PLN-R14del Cardiomyopathy (#3432) Location: MCEC Exhibit and Poster HallPresenting Author: Huanyu Zhou, PhD, Associate Director, Gene Therapy at Tenaya Therapeutics The posters presented at ASGCT will be available in the “Our Science” section of the company’s website at the time of the live presentation. Conference Call and WebcastTenaya management plans to host a webcast conference call to discuss the TN-401 data being presented at the upcoming ASGCT Annual Meeting 2026. Details will be posted to the “Events & Presentations” page in the investor section of the Tenaya website at www.tenayatherapeutics.com. About PKP2-Associated ARVCPlakophilin-2 (PKP2) mutations are the most common genetic cause of arrhythmogenic right ventricular cardiomyopathy (ARVC, also known as arrhythmogenic cardiomyopathy or ACM), occurring in approximately 40 percent of the overall ARVC population. The prevalence of PKP2-associated ARVC is estimated at more than 70,000 people in the U.S. alone. In PKP2-associated ARVC, mutations of the PKP2 gene results in insufficient expression of a protein needed for the proper functioning of the desmosomal complex that maintains physical connections and electrical signaling between heart muscle cells. As the desmosome structure degrades, cardiac muscle cells are replaced by fibrofatty tissue and electrical impulses in the heart become unstable, resulting in irregular heart rhythms that can be fatal. ARVC symptoms include arrhythmias, palpitations, lightheadedness, dizziness and fainting. It is typically diagnosed before age 40, and sudden cardiac arrest due to life-threatening ventricular arrhythmias is frequently the first manifestation of disease. Current treatments include anti-arrhythmic medications, implantable cardioverter-defibrillators (ICDs) and ablation procedures, which do not address the underlying genetic cause of disease. About TN-401 Gene Therapy and the RIDGE-1 Clinical TrialTN-401 is an investigational AAV9-based gene therapy being developed for the treatment of ARVC due to mutations in the PKP2 gene. AAV9 was selected as the vector for delivery of Tenaya’s PKP2 gene therapy based on its extensive clinical and commercial safety record and demonstrated ability to target heart muscle cells. TN-401 has received Orphan Drug and Fast Track Designations from the U.S. Food and Drug Administration. Tenaya’s development of TN-401 is supported in part by a grant from the California Institute for Regenerative Medicines (CIRM). The RIDGE-1 Phase 1b/2 clinical trial of TN-401 in patients with PKP2-associated ARVC is a multi-center, open-label, dose escalation study being conducted in the U.S. and UK. RIDGE-1 is intended to assess the safety, tolerability and preliminary clinical efficacy of a one-time intravenous infusion of TN-401. RIDGE-1 will seek to enroll up to fifteen adults who have been diagnosed with PKP2-associated ARVC, have an ICD and have high counts of premature ventricular contractions (PVCs) during screening, indicating electrical instability and increased risk of fatal arrhythmias. To learn more about gene therapy for ARVC and the RIDGE-1 clinical trial, please visit ARVCstudies.com or ClinicalTrials.gov (NCT06228924). About Tenaya TherapeuticsTenaya Therapeutics is a clinical-stage biotechnology company committed to a bold mission: to discover, develop and deliver potentially curative therapies that address the underlying drivers of heart disease. Tenaya’s pipeline includes clinical-stage candidates TN-201, a gene therapy for MYBPC3-associated hypertrophic cardiomyopathy (HCM) and TN-401, a gene therapy for PKP2-associated arrhythmogenic right ventricular cardiomyopathy (ARVC) and TN-301, a clinical-stage small molecule HDAC6 inhibitor for the potential treatment of heart failure and related cardio/muscular disease, such as Duchenne’s muscular dystrophy. Tenaya has employed a suite of integrated internal capabilities, including modality agnostic target validation, capsid engineering and manufacturing, to generate a portfolio of novel medicines based on genetic insights, including multiple early-stage programs in preclinical development aimed at the treatment of both rare genetic disorders and more prevalent heart conditions. Tenaya is also leveraging this expertise through a research collaboration with Alnylam Pharmaceuticals to discover novel human genetic targets for the potential development of disease-modifying treatments for cardiovascular diseases. For more information, visit www.tenayatherapeutics.com. Forward-Looking StatementsThis press release contains forward-looking statements as that term is defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Statements in this press release that are not purely historical are forward-looking statements. Words such as “planned,” “potential,” “will,” and similar expressions are intended to identify forward-looking statements. Such forward-looking statements include, among other things, the therapeutic potential of TN-401 as a treatment for ARVC caused by mutations in the PKP2 gene; the timing and content of the RIDGE-1 data presentation and related conference call, as well as Tenaya’s other ASGCT poster presentations; and TN-501 as a potential treatment for PLN R14del-associated DCM. The forward-looking statements contained herein are based upon Tenaya’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. These forward-looking statements are neither promises nor guarantees and are subject to a variety of risks and uncertainties, including but not limited to: availability of data at the referenced times; the timing and progress of RIDGE-1 and Tenaya’s other ongoing clinical trials; the potential failure of Tenaya’s product candidates to demonstrate safety and/or efficacy in clinical testing; the potential for any clinical trial results to differ from preclinical, interim, preliminary, topline or expected results; risks associated with the process of discovering, developing and commercializing therapies that are safe and effective for use as human therapeutics; Tenaya’s ability to develop, initiate or complete preclinical studies and clinical trials, and obtain approvals, for any of its product candidates; Tenaya’s continuing compliance with applicable legal and regulatory requirements; Tenaya’s ability to raise any additional funding it will need to continue to pursue its business and product development plans; Tenaya’s reliance on third parties; Tenaya’s manufacturing, commercialization and marketing capabilities and strategy; the loss of key scientific or management personnel; competition in the industry in which Tenaya operates; Tenaya’s ability to obtain and maintain intellectual property protection for its product candidates; general economic and market conditions; and other risks. Information regarding the foregoing and additional risks may be found in the section entitled “Risk Factors” in documents that Tenaya files from time to time with the Securities and Exchange Commission. These forward-looking statements are made as of the date of this press release, and Tenaya assumes no obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. Tenaya ContactsMichelle CorralVP, Corporate Communications and Investor RelationsIR@tenayathera.com InvestorsAnne-Marie FieldsPrecision AQannemarie.fields@precisionaq.com MediaWendy RyanTen Bridge Communicationswendy@tenbridgecommunications.com
Anaxiom Announces Early Clinical Results from First-in-Human Experience of Novel Minimally Invasive Obesity Device; Advances Toward U.S. Early Feasibility Study
LAGUNA HILLS, Calif.–(BUSINESS WIRE)–Anaxiom Corporation, a clinical-stage medical device company developing a novel, non-surgical treatment for obesity, today announced the successful completion of a 12-patient OUS clinical study. Early results demonstrate device safety and promising weight loss supporting advancement toward a U.S. Early Feasibility Study. Promising Early Clinical Experience The […]
Late-Breaking Data from PRESERVE Trial Evaluates XVIVO’s HOPE Preservation Approach in Heart Transplantation
Preliminary results suggest potential to support extended preservation time and donor heart utilization TORONTO–(BUSINESS WIRE)–XVIVO, a global medtech company in the organ transplant industry, today announced late-breaking preliminary data from the PRESERVE Trial (NCT05881278) evaluating hypothermic oxygenated perfusion (HOPE) using the XVIVO Heart Assist Transport™. The trial met its pre-specified […]
Atrium Therapeutics Earns $15 Million Milestone Payment from Bristol Myers Squibb Under Global Cardiovascular Collaboration
SAN DIEGO, April 23, 2026 /PRNewswire/ — Atrium Therapeutics, Inc. (Nasdaq: RNA) (the “Company”), a biopharmaceutical company dedicated to delivering RNA therapeutics directly to the heart, announced today it has earned a $15 million development milestone payment from Bristol Myers…
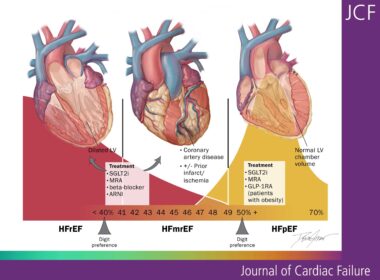
New HFSA Scientific Statement Provides Practical Guidance for Managing Heart Failure with Mildly Reduced Ejection Fraction
New guidance reframes HFmrEF as a distinct, clinically meaningful condition requiring consistent recognition and management WASHINGTON, April 22, 2026 /PRNewswire/ — A new Scientific Statement from the Heart Failure Society of America (HFSA) challenges longstanding ambiguity in the…