SAN FRANCISCO–(BUSINESS WIRE)– #ai–Viz.ai, the leader in AI-powered disease detection and intelligent care coordination, today announced a strategic commercialization collaboration with Johnson & Johnson to expand access to Viz.ai’s Subdural Hemorrhage software solution across hospitals in the United States for automated detection, labeling and quantification of subdural collections. By integrating the Viz Subdural solution into its neurovascular offerings, the company aims to support hospital
Other News
Philips launches new Bridge Plus Occlusion Balloon to help manage rare but life-threatening SVC tears during lead extraction
Clinical illustration of the Philips Bridge Plus balloon in use
Clinical illustration of the Philips Bridge Plus balloon in use
The Philips Bridge Plus Balloon, uninflated prior to use
The Philips Bridge Plus Balloon, uninflated prior to use
The inflated Philips Bridge Plus Balloon
The inflated Philips Bridge Plus Balloon
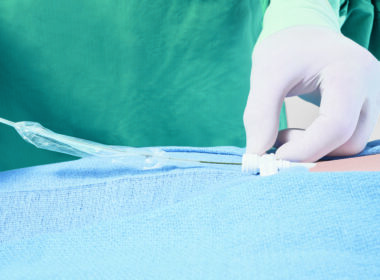
April 23, 2026 Bridge Plus was designed for rare, life-threatening emergencies to help control bleeding during superior vena cava (SVC) tears, which occur in
Nanox Signs Distribution Agreement with Radiology Oncology Systems to Expand Nanox.ARC Adoption in the United States
Expands Nanox’s growing network of U.S. distribution partnerships
Atrium Therapeutics Earns $15 Million Milestone Payment from Bristol Myers Squibb Under Global Cardiovascular Collaboration
SAN DIEGO, April 23, 2026 /PRNewswire/ — Atrium Therapeutics, Inc. (Nasdaq: RNA) (the “Company”), a biopharmaceutical company dedicated to delivering RNA therapeutics directly to the heart, announced today it has earned a $15 million development milestone payment from Bristol Myers…
Conavi Medical Awarded 2026 TAG Innovation Award
Recognition Follows FDA 510(k) Clearance of its Next-Generation Hybrid Intravascular Imaging System Recognition Follows FDA 510(k) Clearance of its Next-Generation Hybrid Intravascular Imaging System
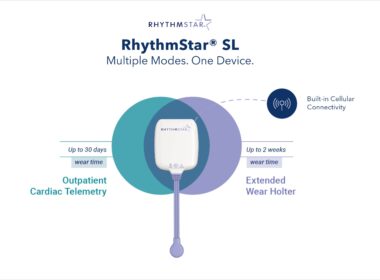
RhythMedix Launches Next-Generation RhythmStar® SL Cardiac Monitor
Advancing Remote Cardiac Monitoring with Faster Insights, Greater Comfort, and Seamless Connectivity MOUNT LAUREL, N.J., April 22, 2026 /PRNewswire/ — RhythMedix, LLC (RhythMedix), a nationwide U.S.-based cardiac monitoring company, today announced the launch of its next-generation…
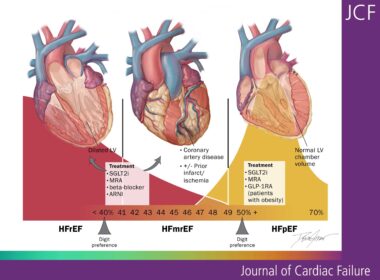
New HFSA Scientific Statement Provides Practical Guidance for Managing Heart Failure with Mildly Reduced Ejection Fraction
New guidance reframes HFmrEF as a distinct, clinically meaningful condition requiring consistent recognition and management WASHINGTON, April 22, 2026 /PRNewswire/ — A new Scientific Statement from the Heart Failure Society of America (HFSA) challenges longstanding ambiguity in the…
Sonorous Neurovascular Receives FDA 510(k) Clearance for BosCATH™ Neurovascular Catheter
LAKE FOREST, Calif., April 22, 2026 /PRNewswire/ — Sonorous Neurovascular, a pioneering medical device company focused on innovative solutions for cerebral venous diseases and neurovascular interventions, today announced that it has received 510(k) clearance from the U.S. Food and Drug…
Microbot Medical® Welcomes Updated Occupational Protection Guidelines from Leading Medical Societies, Strengthening Commercial Position of LIBERTY®
Members of the Society of Interventional Radiology (SIR) and the Cardiovascular and Interventional Radiological Society of Europe (CIRSE) Represent the Largest Addressable User Market for the LIBERTY System At least Nine Medical Societies Endorse Updated Guidelines to Lower Radiation Exposure and Improve Ergonomic Issues Impacting Health Practitioners HINGHAM, Mass., April 22, 2026 (GLOBE NEWSWIRE) — Microbot Medical Inc. (Nasdaq: MBOT), developer and distributor of the innovative LIBERTY® Endovascular Robotic System, today highlighted that the Society of Interventional Radiology (SIR) and the Cardiovascular and Interventional Radiological Society of Europe (CIRSE), recently released updated joint guidelines on occupational protection in interventional radiology. The revised guideline provides important safety guidance to physicians, radiographers, nurses, medical physics experts, and all other staff involved in interventional radiology procedures. The guidelines, which were published in CardioVascular and Interventional Radiology (CVIR), cover updated clinical evidence, new exposure sources and address musculoskeletal risks for interventional radiology staff and, for the first time, include a section on radiation protection during pregnancy for female practitioners. “Part of our mission for the LIBERTY System is to enable physicians to continue improving and saving patients’ lives while protecting their own. LIBERTY’s design aims to reduce radiation exposure and physical strain for physicians and related medical staff, positioning the LIBERTY System as a potential solution to address both the health, and the staffing challenges in interventional care,” commented Harel Gadot, Chairman, CEO & President. “It is very encouraging to see increased emphasis on protecting healthcare providers in the interventional space coming from leading medical societies, such as the American Medical Association (AMA), one of the largest medical associations in the U.S., along with SIR and CIRSE.” Prolonged exposure to radiation may have significant long-term health risks, including cancer, cardiovascular disease, reproductive health effects, and cataracts. These risks have also been cited as contributing to the staffing shortage in the endovascular space, especially in interventional radiology, which currently ranks number two among specialties with highest physician shortages. The burden disproportionately affects women, who often cite radiation exposure and the physical demands of wearing lead aprons as barriers to entering or advancing in the field. The LIBERTY System’s remote operation capabilities enable physicians to work away from the radiation source and in a seated position, potentially lowering musculoskeletal strain while improving workflow efficiency. Along with SIR and CIRSE, occupational protection initiatives are being endorsed by the Society of NeuroInterventional Surgery (SNIS), the Society of Vascular and Interventional Neurology (SVIN), the Society for Cardiovascular Angiography and Interventions (SCAI), American College of Cardiology (ACC), the Alliance for Cardiovascular Professionals (ACVP), the American Society of Echocardiography (ASE), the Heart Rhythm Society (HRS), and Society for Vascular Surgery (SVS). Additionally, in late 2025, the American Medical Association (AMA) adopted a new policy to strengthen protections for healthcare professionals and trainees who may face occupational radiation exposure. The policy emphasizes the importance of continued research into the health effects of cumulative exposure to ionizing radiation, as well as education and training to minimize occupational risk to physicians and patients. About Microbot Medical Microbot Medical Inc. (NASDAQ: MBOT) is a commercial stage medical device company focused on transforming endovascular procedures through advanced robotic technology. Microbot’s LIBERTY® Endovascular Robotic System is the world’s first FDA cleared single-use, remotely operated robotic solution designed for precision, efficiency and safety. Backed by a strong intellectual property portfolio and a commitment to innovation, Microbot is driving the future of endovascular care. Learn more at www.microbotmedical.com and connect on LinkedIn and X. Safe Harbor Statements to future financial and/or operating results, future adoption of products, future growth in research, technology, clinical development, commercialization and potential opportunities for Microbot Medical Inc. and its subsidiaries, along with other statements about the future expectations, beliefs, goals, plans, or prospects expressed by management, constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and the Federal securities laws. Any statements that are not historical fact (including, but not limited to statements that contain words such as “contemplates,” “continues,” “could,” “forecasts,” “intends,” “may,” “might,” “possible,” “potential,” “predicts,” “projects,” “should,” “would,” “will,” “believes,” “plans,” “anticipates,” “expects,” “estimates” and similar expressions) should also be considered to be forward-looking statements, but the absence of these words does not mean that a statement is not forward-looking. Forward-looking statements involve risks and uncertainties, including, without limitation, market conditions, risks inherent in the commercialization of the LIBERTY® Endovascular Robotic System, and in the development of future versions of or applications for the system, uncertainty in the results of regulatory pathways and regulatory approvals, uncertainty resulting from political, social and geopolitical conditions, particularly any changes in personnel or processes or procedures at the FDA and announcements of tariffs on imports into the U.S., disruptions resulting from new and ongoing hostilities between Israel and the Palestinians, Iran and other neighboring countries, and maintenance of intellectual property rights. Additional information on risks facing Microbot Medical® can be found under the heading “Risk Factors” in Microbot Medical’s periodic reports filed with the Securities and Exchange Commission (SEC), which are available on the SEC’s web site at www.sec.gov. Microbot Medical® disclaims any intent or obligation to update these forward-looking statements, except as required by law. Contacts: IR@microbotmedical.comMedia@microbotmedical.com
Kestra Showcases Accelerated Innovation at HRS 2026
Kestra Medical Technologies booth at HRS 2026
Kestra Medical Technologies will showcase its Cardiac Recovery System® platform at Heart Rhythm 2026 (HRS). At Booth 2119, attendees can explore the platform through an immersive multimedia experience, experience the ASSURE® system firsthand, and review clinical data from the ACE-PAS study.
KIRKLAND, Wash., April 22, 2026 (GLOBE NEWSWIRE) — Kestra Medical Technologies, Ltd. (Nasdaq: KMTS), a leading wearable medical device and digital healthcare company, today announced it will exhibit at Heart Rhythm 2026, the annual meeting of the Heart Rhythm Society (HRS) taking place April 23–26 in Chicago. The company will showcase its Cardiac Recovery System® platform—a modern, integrated approach to managing sudden cardiac arrest (SCA) risk that unites proven therapy, actionable clinical insights, and ongoing patient support to enhance protection and optimize care. Kestra, one of the fastest-growing companies in medical technology, continues to build momentum as clinicians increasingly shift toward more connected approaches to SCA risk management. At HRS, attendees will see why more clinicians are turning to Kestra to improve patient protection and better manage risk during the most vulnerable stages of recovery. The ASSURE® WCD is at the center of the Cardiac Recovery System platform. Insights from the ASSURE WCD Clinical Evaluation Post-Approval Study (ACE-PAS)—the largest prospective study of wearable defibrillators to date—demonstrate robust real-world performance and continue to guide innovation across the platform. At HRS, Kestra is launching a recently FDA-approved ASSURE WCD algorithm update that builds on market-leading clinical performance and applies real-world data to further enhance patient protection and advance care. “At Kestra, innovation is a core principle, so we are excited to further advance the clinical benefits of the ASSURE WCD with this new algorithm. We’re seeing a clear shift in how clinicians approach SCA risk, with a growing emphasis on patient compliance and connected models of care,” said Brian Webster, President and Chief Executive Officer. “HRS is a pivotal moment to engage with prescribers and demonstrate how our clinically informed innovation is driving that momentum—delivering meaningful improvements for clinicians and their patients.” Kestra will be located at Booth 2119, where attendees can experience the ASSURE system firsthand, explore the Cardiac Recovery System platform through an immersive multimedia experience, and review clinical data from ACE-PAS. About Kestra Kestra Medical Technologies, Ltd. is a leading wearable medical device and digital healthcare company focused on transforming patient outcomes in cardiovascular disease using monitoring and therapeutic intervention technologies that are intuitive, intelligent, and connected. For more information, please visit www.kestramedical.com. Forward-Looking StatementsExcept where otherwise noted, the information contained in this press release is as of April 22, 2026. Statements in this press release that express a belief, expectation or intention, as well as those that are not historical fact, are forward-looking statements. Except as required by law, we undertake no obligation to publicly update any forward-looking statements, whether as a result of new information, future events or otherwise. This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Given their forward-looking nature, these statements involve substantial risks, uncertainties and potentially inaccurate assumptions, and we cannot ensure that any outcome expressed in these forward-looking statements will be realized in whole or in part. You can identify these statements by the fact that they use future dates or use words such as “will,” “may,” “could,” “likely,” “ongoing,” “anticipate,” “estimate,” “expect,” “project,” “intend,” “plan,” “believe,” “assume,” “target,” “forecast,” “guidance,” “goal,” “objective,” “aim,” “seek,” “potential,” “hope” and other words and terms of similar meaning. Among the factors that could cause actual results to differ materially from past results and future plans and projected future results are the following: risks related to our limited operating history and history of net losses; our ability to successfully achieve substantial market adoption of our products; competitive pressures; our ability to adapt our manufacturing and production capacities to evolving patterns of demand, governmental actions and customer trends; product defects or complaints and related liability; our ability to obtain and maintain adequate coverage and reimbursement levels for our products; our ability to comply with changing laws and regulatory requirements and resulting costs; our dependence on a limited number of suppliers; and other risks and uncertainties, including those described under the heading “Risk Factors” in Kestra’s Annual Report on Form 10-K for the fiscal year ended April 30, 2025 filed with the U.S. Securities and Exchange Commission (“SEC”) on July 17, 2025, and in other periodic reports filed by Kestra with the SEC. These filings are available on the Investor Relations section of our website at https://investors.kestramedical.com/ and on the SEC’s website at https://sec.gov/. A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/93288f45-4a54-4de1-b66e-207047603f80 CONTACT: Media contact
Rhiannon Pickus
rhiannon.pickus@kestramedical.com
Investor contact
Neil Bhalodkar
neil.bhalodkar@kestramedical.com